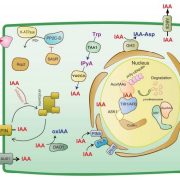
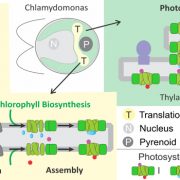
Imaging of Light Absorption and Photosynthesis
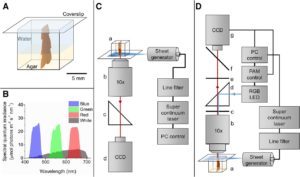 Due to its ease of use and noninvasive nature, variable chlorophyll fluorescence techniques have become increasingly popular for estimating photosynthetic parameters. Most measurements of variable chlorophyll fluorescence in complex plant tissues and surface-associated cell assemblages (e.g. biofilms) rely on external measurements with fiber optic or imaging fluorimeters. Such studies assume that different cells in the sample are subjected to the same amount of measuring light and actinic irradiance, that saturating pulses are indeed saturating all the cells, and that the fluorescence detected is emitted equally from all sampled cells. These assumptions are not met in optically dense samples. Steep light gradients in densely pigmented tissues or algal biofilms distort the measurements of maximal and effective PSII quantum yields. Cells located deeper inside tissues (or biofilms) receive less actinic irradiance than cells close to the surface. Thus, externally integrated measurements of variable chlorophyll fluorescence contain a complex mixture of signals originating from different layers in the structure exposed to different levels of measuring and actinic light, and the actual operational depth of such measurements remains unknown. These inherent limitations can lead to overestimations of effective PSII quantum yields. Lichtenberg et al. ( 10.1104/pp.17.00820) have developed a novel system that allows a comparison of how measured distributions of PSII quantum yields in plant tissue change under natural tissue light gradients compared with conventionally measured quantum yields with uniform exposures to actinic light. This was achieved by applying actinic irradiance perpendicular to one side of thallus cross sections of the aquatic macrophyte Fucus vesiculosus with laser light sheets of defined spectral composition, while imaging variable chlorophyll fluorescence from cross sections with a microscope-mounted pulse amplitude-modulated imaging system. The authors show that quantum yields are highly affected by light gradients and that traditional surface-based variable chlorophyll fluorescence measurements result in substantial underestimations and/or overestimations, depending on incident actinic irradiance.
Due to its ease of use and noninvasive nature, variable chlorophyll fluorescence techniques have become increasingly popular for estimating photosynthetic parameters. Most measurements of variable chlorophyll fluorescence in complex plant tissues and surface-associated cell assemblages (e.g. biofilms) rely on external measurements with fiber optic or imaging fluorimeters. Such studies assume that different cells in the sample are subjected to the same amount of measuring light and actinic irradiance, that saturating pulses are indeed saturating all the cells, and that the fluorescence detected is emitted equally from all sampled cells. These assumptions are not met in optically dense samples. Steep light gradients in densely pigmented tissues or algal biofilms distort the measurements of maximal and effective PSII quantum yields. Cells located deeper inside tissues (or biofilms) receive less actinic irradiance than cells close to the surface. Thus, externally integrated measurements of variable chlorophyll fluorescence contain a complex mixture of signals originating from different layers in the structure exposed to different levels of measuring and actinic light, and the actual operational depth of such measurements remains unknown. These inherent limitations can lead to overestimations of effective PSII quantum yields. Lichtenberg et al. ( 10.1104/pp.17.00820) have developed a novel system that allows a comparison of how measured distributions of PSII quantum yields in plant tissue change under natural tissue light gradients compared with conventionally measured quantum yields with uniform exposures to actinic light. This was achieved by applying actinic irradiance perpendicular to one side of thallus cross sections of the aquatic macrophyte Fucus vesiculosus with laser light sheets of defined spectral composition, while imaging variable chlorophyll fluorescence from cross sections with a microscope-mounted pulse amplitude-modulated imaging system. The authors show that quantum yields are highly affected by light gradients and that traditional surface-based variable chlorophyll fluorescence measurements result in substantial underestimations and/or overestimations, depending on incident actinic irradiance.