Suc Signals Induce Etiolated Stem Branching
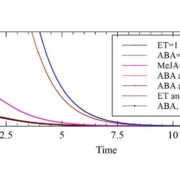
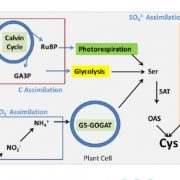
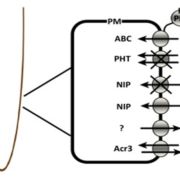
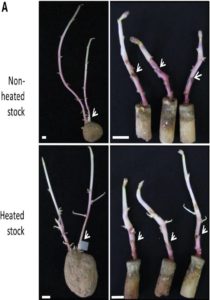 Plant shoot branching is determined by apical dominance, a process in which the apical bud (shoot tip) inhibits the outgrowth of axillary buds further down the stem to control the number of growing branches. In response to this inhibition, plants have evolved rapid long-distance signaling mechanisms to release axillary buds and replenish the plant with new growing shoot tips. The nutrition hypothesis of apical dominance assumes that access to plant nutrients is the major factor regulating axillary bud growth. In particular, it has been suggested apical dominance is maintained largely by the sugar demand of the shoot tip, which limits the amount of sugar available to the axillary buds. Small-molecule sugars, however, are also known to act as signaling molecules in many physiological processes. Long cold storage induces loss of potato (Solanum tuberosum) tuber apical dominance and results in secondary branching. Salam et al. (pp. 10.1104/pp.17.00995 ) now show that a similar branching pattern can be induced by short heat treatment of the tubers. Grafting experiments showed that the scion branches only when grafted onto heat- or cold-treated tuber parenchyma, suggesting that the branching signal is transmitted systemically. The exogenous supply of Suc, Glc, or Fru solutions to detached sprouts induced branching in a dose-responsive manner, and an increase in Suc level was observed in tuber parenchyma upon branching induction, suggesting a role for elevated parenchyma sugars in the regulation of branching. The authors also demonstrate that by silencing the gene encoding vacuolar invertase in transgenic potato plants, they can induce enhanced branching of the tuber. However, sugar analysis of the apex and node after grafting showed no distinct differences in sugar levels between branching and nonbranching stems. These results suggest that Suc in the tuber parenchyma induces branching not by excess mobilization from the parenchyma to the stem but through long-distance signaling.
Plant shoot branching is determined by apical dominance, a process in which the apical bud (shoot tip) inhibits the outgrowth of axillary buds further down the stem to control the number of growing branches. In response to this inhibition, plants have evolved rapid long-distance signaling mechanisms to release axillary buds and replenish the plant with new growing shoot tips. The nutrition hypothesis of apical dominance assumes that access to plant nutrients is the major factor regulating axillary bud growth. In particular, it has been suggested apical dominance is maintained largely by the sugar demand of the shoot tip, which limits the amount of sugar available to the axillary buds. Small-molecule sugars, however, are also known to act as signaling molecules in many physiological processes. Long cold storage induces loss of potato (Solanum tuberosum) tuber apical dominance and results in secondary branching. Salam et al. (pp. 10.1104/pp.17.00995 ) now show that a similar branching pattern can be induced by short heat treatment of the tubers. Grafting experiments showed that the scion branches only when grafted onto heat- or cold-treated tuber parenchyma, suggesting that the branching signal is transmitted systemically. The exogenous supply of Suc, Glc, or Fru solutions to detached sprouts induced branching in a dose-responsive manner, and an increase in Suc level was observed in tuber parenchyma upon branching induction, suggesting a role for elevated parenchyma sugars in the regulation of branching. The authors also demonstrate that by silencing the gene encoding vacuolar invertase in transgenic potato plants, they can induce enhanced branching of the tuber. However, sugar analysis of the apex and node after grafting showed no distinct differences in sugar levels between branching and nonbranching stems. These results suggest that Suc in the tuber parenchyma induces branching not by excess mobilization from the parenchyma to the stem but through long-distance signaling.