CRISPR Mutants Shed Light on Pectin’s Role in Tomato Fruit Softening
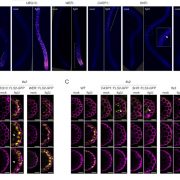
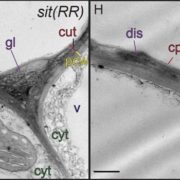
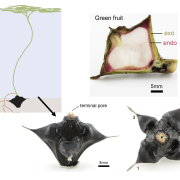
 Tomato (Solanum lycopersicum) fruits undergo pronounced softening during ripening. Softening is important for flavor development and overall palatability, but also impacts fruit storage, transportability, and shelf life. Shelf life is a particularly important quality trait of tomato fruits affected by alterations in the cuticle properties of the fruits and the remodeling of fruit cell walls. Studies with transgenic tomato plants have indicated that a range of pectin-degrading enzymes are involved in fruit softening by way of cell wall remodeling. Until recently, pectin was thought to contribute to wall mechanics independently of other cell wall polymers such as cellulose and xyloglucan. However, the validity of this conventional role of pectin has been challenged by a series of recent discoveries which support the idea that pectin may directly contribute to the cross linking of cellulose microfibrils in the cell wall, potentially to a greater extent than xyloglucan, the classical cross-linking hemicellulose. Previous molecular studies of ripening-related genes have usually involved silencing of only a single gene and it has proved difficult to compare the effects of silencing these genes across the different experimental systems. Wang et al. (10.1104/pp.18.01187) now report the generation of CRISPR-based mutants in the ripening-related genes encoding the pectin-degrading enzymes pectate lyase (PL), polygalacturonase 2a (PG2a), and β-galactanase (TBG4). Comparison of the physiochemical properties of the fruits from a range of PL, PG2a, and TBG4 CRISPR lines demonstrated that only mutations in PL resulted in firmer fruits, although mutations in PG2a and TBG4 influenced fruit color and weight. The presence of all three enzyme activities, however, are needed to allow normal ripening-related changes in pericarp cell-to-cell adhesion and solubilization of pectin from association with cellulose microfibrils. Pectin localization, distribution, and solubility in the pericarp cells of the CRISPR mutant fruits were also investigated. In toto, the data obtained indicate that PL, PG2a, and TBG4 act on separate cell wall domains.
Tomato (Solanum lycopersicum) fruits undergo pronounced softening during ripening. Softening is important for flavor development and overall palatability, but also impacts fruit storage, transportability, and shelf life. Shelf life is a particularly important quality trait of tomato fruits affected by alterations in the cuticle properties of the fruits and the remodeling of fruit cell walls. Studies with transgenic tomato plants have indicated that a range of pectin-degrading enzymes are involved in fruit softening by way of cell wall remodeling. Until recently, pectin was thought to contribute to wall mechanics independently of other cell wall polymers such as cellulose and xyloglucan. However, the validity of this conventional role of pectin has been challenged by a series of recent discoveries which support the idea that pectin may directly contribute to the cross linking of cellulose microfibrils in the cell wall, potentially to a greater extent than xyloglucan, the classical cross-linking hemicellulose. Previous molecular studies of ripening-related genes have usually involved silencing of only a single gene and it has proved difficult to compare the effects of silencing these genes across the different experimental systems. Wang et al. (10.1104/pp.18.01187) now report the generation of CRISPR-based mutants in the ripening-related genes encoding the pectin-degrading enzymes pectate lyase (PL), polygalacturonase 2a (PG2a), and β-galactanase (TBG4). Comparison of the physiochemical properties of the fruits from a range of PL, PG2a, and TBG4 CRISPR lines demonstrated that only mutations in PL resulted in firmer fruits, although mutations in PG2a and TBG4 influenced fruit color and weight. The presence of all three enzyme activities, however, are needed to allow normal ripening-related changes in pericarp cell-to-cell adhesion and solubilization of pectin from association with cellulose microfibrils. Pectin localization, distribution, and solubility in the pericarp cells of the CRISPR mutant fruits were also investigated. In toto, the data obtained indicate that PL, PG2a, and TBG4 act on separate cell wall domains.