True Blue: How Cry1 Inhibits Phototropism in Green Seedlings
Kasper van Gelderen
Plant Ecophysiology, Dept. of Biology, Utrecht University, Padualaan 8 3584CH, Utrecht, The Netherlands.
The study of plant movement towards light, also called phototropism, has a venerable history with giants such as Charles Darwin and Frits Went involved in its study, which led to the discovery of the plant hormone auxin (Went, 1928). We now have a deep understanding of its mechanistic underpinnings (Christie and Murphy, 2013). Therefore, it is all the more surprising that fundamentals of phototropic responses can still be uncovered. Among these discoveries, Ballaré et al. (1992) observed that phototropism is enhanced when plants are growing within a canopy. Plants sense the crowding under a canopy by a lowering of the ratio of far-red (FR) to red (R) light (low R:FR; LRFR). This effect arises from the enhanced reflection of FR-light from neighboring leaves. The surrounding leaves also filter and reduce the available blue light (low blue light; LBL). LRFR and LBL promote stem elongation of plants and this competitive response is seen as an adaptive change to accelerate plant growth out of the canopy and reach sunlight, the main resource of plant life. So, which of these light cues is important and which photoreceptors are involved?
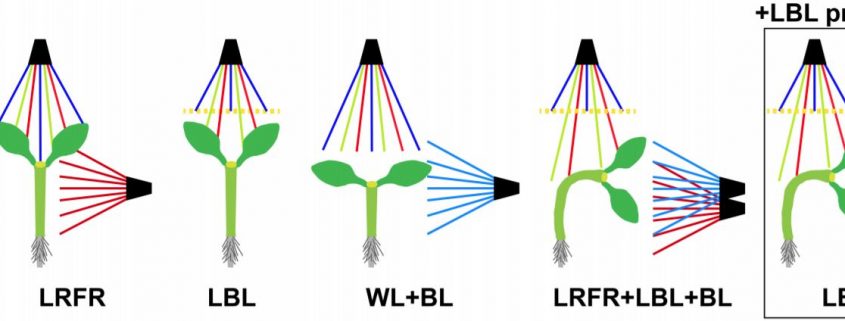
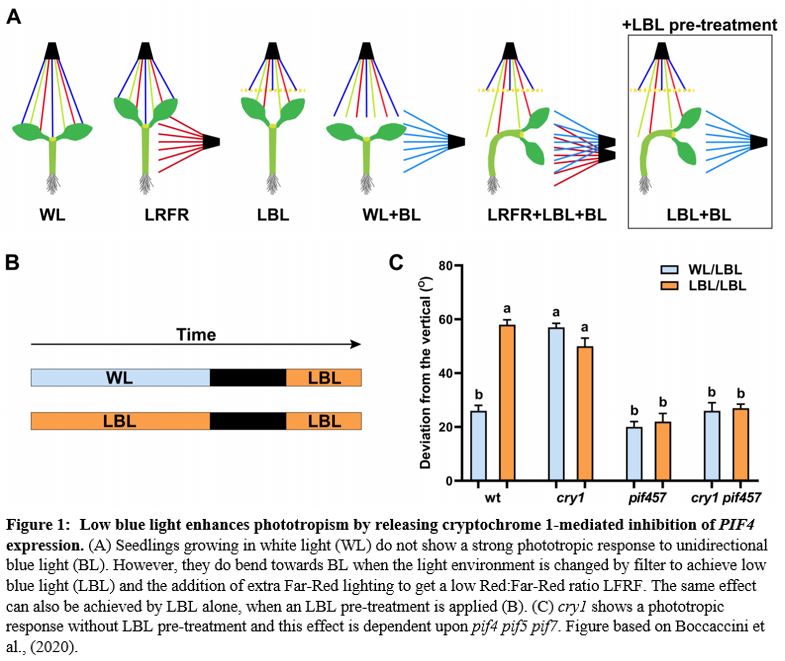 In the previous issue of Plant Physiology, Boccaccini et al. (2020) showed that in Arabidopsis thaliana, a combination of LBL and LRFR promotes phototropism, and that LBL alone is sufficient. In addition, they demonstrate that the blue light receptor cryptochrome 1 (cry1) is active in this response. Previous work had shown that LRFR can promote phototropism in artificial lighting conditions (Goyal et al., 2016), where etiolated seedlings are exposed to a unidirectional source of blue light. However, with a more natural lighting regime, Boccaccini et al. found that LRFR was not sufficient. Only a combination of LBL and LRFR resulted in increased phototropism towards a blue light source within the white growth light (WL, Figure 1A). A pre-treatment of LBL combined with LBL during the phototropic response led to the same result as the combined LBL and LRFR treatment. Thus, the authors proceeded to further investigate this LBL-mediated mechanism by comparing the LBL condition with a low blue pre-treatment (LBL/LBL) and without (WL/LBL) (Figure 1B).
In the previous issue of Plant Physiology, Boccaccini et al. (2020) showed that in Arabidopsis thaliana, a combination of LBL and LRFR promotes phototropism, and that LBL alone is sufficient. In addition, they demonstrate that the blue light receptor cryptochrome 1 (cry1) is active in this response. Previous work had shown that LRFR can promote phototropism in artificial lighting conditions (Goyal et al., 2016), where etiolated seedlings are exposed to a unidirectional source of blue light. However, with a more natural lighting regime, Boccaccini et al. found that LRFR was not sufficient. Only a combination of LBL and LRFR resulted in increased phototropism towards a blue light source within the white growth light (WL, Figure 1A). A pre-treatment of LBL combined with LBL during the phototropic response led to the same result as the combined LBL and LRFR treatment. Thus, the authors proceeded to further investigate this LBL-mediated mechanism by comparing the LBL condition with a low blue pre-treatment (LBL/LBL) and without (WL/LBL) (Figure 1B).
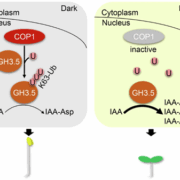
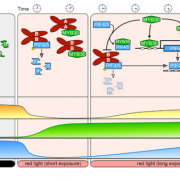
The main photoreceptors regulating phototropism are the phototropins (phot), and in a phot1 phot2 double knockout no phototropism is observed. However, plants have another set of blue-light photoreceptors, the cryptochromes. Boccaccini et al discovered that the cry1 mutant has an increased phototropism response in WL/LBL, without a further increase in bending in the LBL/LBL condition, compared to wild type, which only had a strong response with the low blue pre-treatment (LBL/LBL, Figure 1C). This result indicates that cry1 normally inhibits the phototropism response in a low blue environment. However, an increase in blue light from one side can deactivate cry1 and therefore allow phototropism to occur. The most important downstream actors of the cry and phytochrome photoreceptors are the PHYTOCHROME INTERACTING FACTORS (PIFs). The triple mutant, pif4, pif5 and pif7 is lacking in phototropic bending. This pif4 pif5 pif7 mutant, when crossed with cry1, still does not have the phototropic response, showing that these PIFs are necessary for cry1 to inhibit phototropism (Figure 1C). During a typical day, PIF4 and PIF5 levels gradually decrease; however the authors found that the LBL/LBL treatment causes PIF levels to remain higher for longer. This accumulation of PIF4 depends upon a functional cry1. Overexpressing lines of PIF4 and PIF5 had enhanced phototropism responses, showing that increases in PIF4/5 levels are connected to phototropism. In conclusion, the authors convincingly show that cry1 inhibits PIF4/5 and that the unidirectional blue light allows a buildup of PIFs. Of course, one asks: how does a buildup of PIFs lead to a directional response? From previous research we know that PIFs activate auxin biosynthesis and transport during phototropism (Goyal et al., 2016). Therefore, the authors also show that in their setup an auxin gradient builds up away from the blue light source and that this is dependent upon the PIN-FORMED auxin efflux carriers PIN3,4 and 7, which, during phototropism, pump auxin out from the vasculature tissue, towards the cortex and epidermis (Ding et al. 2011).
In short, this paper bridges a long-standing gap between traditional phototropism studies that often use highly artificial lighting regimes and etiolated seedlings exposed to unidirectional monochromatic light. Boccaccini et al. combine these studies with work using green seedlings and growth under regimes with blue light enrichment (Figure 1A), and they relate these studies to the real world in which a growing plant growing often experiences canopy shading. In the latter case, the authors even grow Arabidopsis in a field, next to a grass covering, to show that both cry1 and cry1 pif457 mutants behave the same as in the lab. Altogether, the findings in this paper neatly shows how plants use competition and shading cues to grow towards the sun and enhance their future success.
References
Ballaré, C.L., Scopel, A.L., Radosevich, S.R., and Kendrick, R.E. (1992). Phytochrome-mediated phototropism in de-etiolated seedlings: Occurrence and ecological significance. Plant Physiol. 100: 170–177.
Christie, J.M. and Murphy, A.S. (2013). Shoot phototropism in higher plants: New light through old concepts. Am. J. Bot. 100: 35–46.
Ding, Z., Galván-Ampudia, C.S., Demarsy, E., Langowski, L., Kleine-Vehn, J., Fan, Y., Morita, M.T., Tasaka, M., Fankhauser, C., Offringa, R., Friml, J., and Łangowski, Ł. (2011). Light-mediated polarization of the PIN3 auxin transporter for the phototropic response in Arabidopsis. Nat. Cell Biol. 13: 447–52.
Goyal, A., Karayekov, E., Galvão, V.C.C., Ren, H., Casal, J.J.J., and Fankhauser, C. (2016). Shade Promotes Phototropism through Phytochrome B-Controlled Auxin Production. Curr. Biol. 26: 1–8.
Went, F.W. (1928). Wuchsstoff und Wachstum. Rec. Trav. Bot. Neerl. 25: 1–116.