Folate, DNA Methylation and Flowering Time
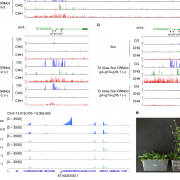
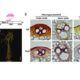
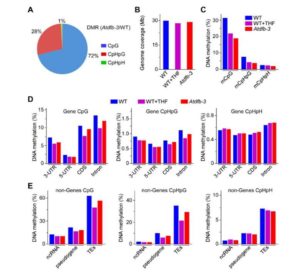 Tetrahydrofolate (THF) and its derivatives, collectively termed folates, are a group of essential B-complex vitamins that have long been recognized as necessary nutrients to support normal cell differentiation and growth. Folates function as co-enzymes in one-carbon transfer reactions and play a central role in synthesis of nucleotides and amino acids. Dysfunction of cellular folate metabolism leads to serious defects in plant development; however, the molecular mechanisms of folate-mediated cellular modifications and physiological responses in plants are still largely unclear. Wang et al. () now report that THF controls Arabidopsis thaliana flowering time by adjusting DNA methylation-regulated gene expression. Changes in chromatin structure due to altered DNA methylation patterns have previously been implicated in the transition to flowering. One gene in particular, FWA (Flowering Wageningen), has been extensively studied since it was originally characterized from epigenetic mutants displaying a heritable late-flowering phenotype. To initiate normal flowering process, FWA expression is required to be silenced by methyltransferase1 (MET1)-mediated cytosine methylation to release its interfering effects on the function of the key flower promoting gene FT (FLOWERING LOCUS T). The authors report that seedlings supplied with THF, as well as the high endogenous Atdfb (dihydrofolate synthetase folypolyglutamate synthetase homolog B) showed dramatically induced effects on the release of MET1-mediated chromatin silencing and gene expression activity in a genome-wide scale, including the flowering regulation gene FWA. With elevated folate conditions, FWA constantly displayed transcription activity leading to retarded floral transition in a dose-dependent manner. Moreover, the loss of function of MET1 dramatically impaired THF-modulated flowering responses. These studies reveal a fundamental role of folate homeostasis in epigenetically controlled gene expression.
Tetrahydrofolate (THF) and its derivatives, collectively termed folates, are a group of essential B-complex vitamins that have long been recognized as necessary nutrients to support normal cell differentiation and growth. Folates function as co-enzymes in one-carbon transfer reactions and play a central role in synthesis of nucleotides and amino acids. Dysfunction of cellular folate metabolism leads to serious defects in plant development; however, the molecular mechanisms of folate-mediated cellular modifications and physiological responses in plants are still largely unclear. Wang et al. () now report that THF controls Arabidopsis thaliana flowering time by adjusting DNA methylation-regulated gene expression. Changes in chromatin structure due to altered DNA methylation patterns have previously been implicated in the transition to flowering. One gene in particular, FWA (Flowering Wageningen), has been extensively studied since it was originally characterized from epigenetic mutants displaying a heritable late-flowering phenotype. To initiate normal flowering process, FWA expression is required to be silenced by methyltransferase1 (MET1)-mediated cytosine methylation to release its interfering effects on the function of the key flower promoting gene FT (FLOWERING LOCUS T). The authors report that seedlings supplied with THF, as well as the high endogenous Atdfb (dihydrofolate synthetase folypolyglutamate synthetase homolog B) showed dramatically induced effects on the release of MET1-mediated chromatin silencing and gene expression activity in a genome-wide scale, including the flowering regulation gene FWA. With elevated folate conditions, FWA constantly displayed transcription activity leading to retarded floral transition in a dose-dependent manner. Moreover, the loss of function of MET1 dramatically impaired THF-modulated flowering responses. These studies reveal a fundamental role of folate homeostasis in epigenetically controlled gene expression.







Leave a Reply
Want to join the discussion?Feel free to contribute!