A Possible Strategy for Increasing Methionine Titer in Seeds
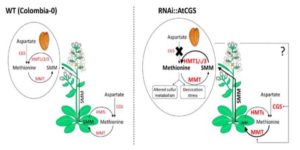 Methionine is a nutritionally essential sulfur-containing amino acid found at low levels in plants and in their seeds. It often limits the nutritional value of crop plants as a source of dietary protein for humans and animals. In plants, methionine plays key roles in protein synthesis and mRNA translation, and regulates indirectly a variety of cellular processes through its main catabolic product S-adenosylmethionine (SAM). SAM serves as the precursor for the synthesis of the plant hormone ethylene, polyamines and biotin, and donates a primary methyl group that is essential for methylation reactions involved in a variety of developmental processes in plant cells. Genetic and biochemical studies suggest that in seeds, methionine can be synthesized de novo as in vegetative tissues via the classical aspartate family pathway by the activity of its main regulatory enzyme, CYSTATHIONINE γ-SYNTHASE (CGS). However, isotope-labeling experiments suggest that methionine can be synthesized in seeds through an alternative pathway by which methionine produced in vegetative tissues is converted to S-methylmethionine (SMM) that is then transported via the phloem into the reproductive tissues where it is converted to methionine. Cohen et al. () have previously produced transgenic Arabidopsis RNAi seeds with lower transcript expression of CYSTATHIONINE γ- SYNTHASE (AtCGS) and found unexpectedly that the seeds accumulated significantly higher levels of methionine compared to controls. Using radiolabels, the authors now show that SMM synthesized in the rosette leaves of the RNAi plants significantly contributed to the accumulation of methionine in their seeds at late stages of development. Seed-specific repression of AtCGS in RNAi seeds triggered the induction of genes operating in the SMM cycle of rosette leaves leading to elevated transport of SMM towards the seeds, where higher reconversion rates of SMM-to-methionine were detected. The results of this study suggest new strategies for improving methionine contents in seeds.
Methionine is a nutritionally essential sulfur-containing amino acid found at low levels in plants and in their seeds. It often limits the nutritional value of crop plants as a source of dietary protein for humans and animals. In plants, methionine plays key roles in protein synthesis and mRNA translation, and regulates indirectly a variety of cellular processes through its main catabolic product S-adenosylmethionine (SAM). SAM serves as the precursor for the synthesis of the plant hormone ethylene, polyamines and biotin, and donates a primary methyl group that is essential for methylation reactions involved in a variety of developmental processes in plant cells. Genetic and biochemical studies suggest that in seeds, methionine can be synthesized de novo as in vegetative tissues via the classical aspartate family pathway by the activity of its main regulatory enzyme, CYSTATHIONINE γ-SYNTHASE (CGS). However, isotope-labeling experiments suggest that methionine can be synthesized in seeds through an alternative pathway by which methionine produced in vegetative tissues is converted to S-methylmethionine (SMM) that is then transported via the phloem into the reproductive tissues where it is converted to methionine. Cohen et al. () have previously produced transgenic Arabidopsis RNAi seeds with lower transcript expression of CYSTATHIONINE γ- SYNTHASE (AtCGS) and found unexpectedly that the seeds accumulated significantly higher levels of methionine compared to controls. Using radiolabels, the authors now show that SMM synthesized in the rosette leaves of the RNAi plants significantly contributed to the accumulation of methionine in their seeds at late stages of development. Seed-specific repression of AtCGS in RNAi seeds triggered the induction of genes operating in the SMM cycle of rosette leaves leading to elevated transport of SMM towards the seeds, where higher reconversion rates of SMM-to-methionine were detected. The results of this study suggest new strategies for improving methionine contents in seeds.







Leave a Reply
Want to join the discussion?Feel free to contribute!