Update: Origins and evolution of stomatal development
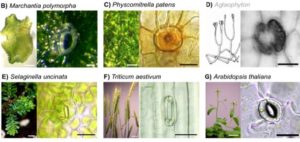 The fossil record suggests stomata-like pores were present on the surfaces of land plants over 400 million years ago. Whether stomata arose once or whether they arose independently across newly evolving land plant lineages has long been a matter of debate. In Arabidopsis, a genetic toolbox has been identified which tightly controls stomatal development and patterning. This includes the bHLH transcription factors SPEECHLESS, MUTE, FAMA and ICE/SCREAMs (SCRMs) which promote stomatal formation. These factors are regulated via a signalling cascade which includes mobile EPIDERMAL PATTERNING FACTOR (EPF) peptides to enforce stomatal spacing. Mosses and hornworts, the most ancient extant lineages to possess stomata, possess orthologues of these Arabidopsis stomatal toolbox genes and manipulation in the model bryophyte Physcomitrella patens has shown that the bHLH and EPF components are also required for moss stomatal development and patterning. This supports an ancient and tightly conserved genetic origin of stomata. Here, we review recent discoveries and, by interrogating newly available plant genomes, we advance the story of stomatal development and patterning across land plant evolution. Furthermore, we identify potential orthologues of the key toolbox genes in a hornwort, further supporting a single ancient genetic origin of stomata in the ancestor to all stomatous land plants.
The fossil record suggests stomata-like pores were present on the surfaces of land plants over 400 million years ago. Whether stomata arose once or whether they arose independently across newly evolving land plant lineages has long been a matter of debate. In Arabidopsis, a genetic toolbox has been identified which tightly controls stomatal development and patterning. This includes the bHLH transcription factors SPEECHLESS, MUTE, FAMA and ICE/SCREAMs (SCRMs) which promote stomatal formation. These factors are regulated via a signalling cascade which includes mobile EPIDERMAL PATTERNING FACTOR (EPF) peptides to enforce stomatal spacing. Mosses and hornworts, the most ancient extant lineages to possess stomata, possess orthologues of these Arabidopsis stomatal toolbox genes and manipulation in the model bryophyte Physcomitrella patens has shown that the bHLH and EPF components are also required for moss stomatal development and patterning. This supports an ancient and tightly conserved genetic origin of stomata. Here, we review recent discoveries and, by interrogating newly available plant genomes, we advance the story of stomatal development and patterning across land plant evolution. Furthermore, we identify potential orthologues of the key toolbox genes in a hornwort, further supporting a single ancient genetic origin of stomata in the ancestor to all stomatous land plants.





Leave a Reply
Want to join the discussion?Feel free to contribute!