Ligands and regulatory properties of the HD-ZIPIII START domain
Husbands et al. identify ligands and regulatory properties of the HD-ZIPIII START domain.
By By Aman Y. Husbands (University of Pennsylvania) and Marja C.P. Timmermans (University of Tuebingen).
Background: Development has been compared to a ball rolling down a hill. Cells initially have broad potential, but over time, they make decisions based on local cues which set them down a path to differentiation. Along this trajectory, thousands of genes need to be turned off or on in a carefully choreographed manner to ensure specialized identity. This is accomplished in part by transcription factors whose activities are precisely controlled by inputs operating across multiple regulatory levels. Here, we study the regulation of CLASS III HOMEODOMAIN LEUCINE ZIPPER (HD-ZIPIII) proteins, a 725-million-year-old family of transcription factors that were redeployed throughout evolution to impact nearly all aspects of plant development.
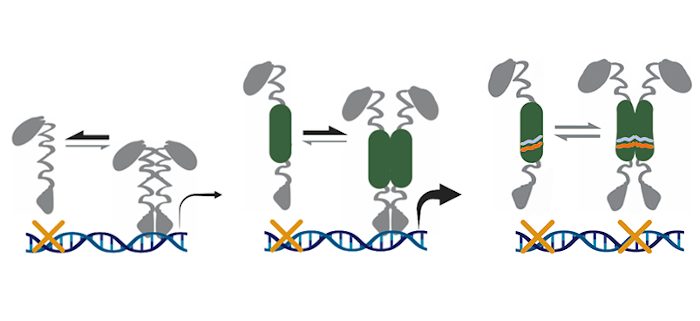
Question: HD-ZIPIII proteins contain a START domain, an evolutionarily ubiquitous module that binds lipophilic ligands. The hypothesis thus arose that HD-ZIPIII activity may be controlled by lipid ligand inputs. Remarkably, both the ligands and regulatory properties of the HD-ZIPIII START domain have remained unknown for over twenty years. The goal of our study was to resolve this long-standing mystery.
Findings: We find that the START domain promotes HD-ZIPIII dimerization and increases their transcriptional potency. We also identify several phospholipid ligands bound by the HD-ZIPIII START domain, and show that perturbing ligand binding abolishes DNA-binding competence. Thus, the START domain turns HD-ZIPIII proteins into potent DNA-binding competent transcription factors but only if they can bind and respond to their phospholipid ligands. Our findings further highlight the remarkably flexible and diverse regulatory potential of START domains.
Next steps: Having established START regulatory properties for an HD-ZIPIII protein in Arabidopsis, we are now considering our findings from an evolutionary perspective. How and when did these regulatory properties come about? Is there functional divergence of START regulation of HD-ZIPIII activity? And how are START domains able to employ such a diverse set of regulatory mechanisms?
Reference:
Aman Y. Husbands, Antje Feller, Vasudha Aggarwal, Courtney E. Dresden, Ashton S. Holub, Taekjip Ha, Marja C.P. Timmermans. (2023). The START domain potentiates HD-ZIPIII transcriptional activity. https://doi.org/10.1093/plcell/koad058