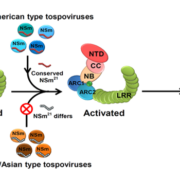
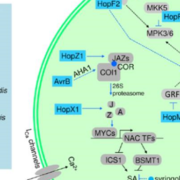
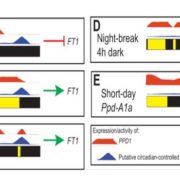
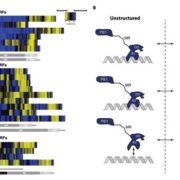
Sensory Plastids: a Novel Form of Specialized Plastid
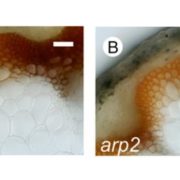
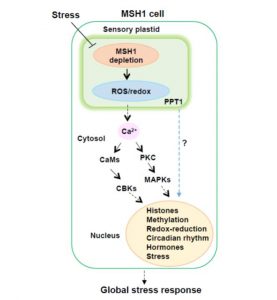 Plastids differentiate into subtypes. For example, the transition of proplastids to chloroplasts defines the photosynthetic tissues within a plant, whereas amyloplasts are formed in nonphotosynthetic tissues. Recently, evidence has accumulated for a novel class of plastids that is specialized for stress sensing and acclimation. These “sensory plastids”, some of which occur in the epidermis, are approximately 30% the size of mesophyll chloroplasts, and are associated with shade and osmotic stress responses. Similar plastids within the vascular parenchyma have been associated with ABA biosynthesis, and high light and drought responses. Beltrán et al. (10.1104/pp.18.00804) reveal that the distinctive proteome of sensory plastids in Arabidopsis (Arabidopsis thaliana) derives from the spatiotemporal regulation of nuclear genes encoding plastid-targeted proteins. In particular, MutS HOMOLOG 1 (MSH1), a plant-specific mitochondrial protein is associated with this specialized plastid type. The global stress phenotype that derives from MSH1 RNAi knockdown is characterized by genome-wide DNA methylome repatterning, implying an epigenomic component to the phenotype. Investigation of the translatome within sensory plastid-containing cells, following MSH1 disruption, provides evidence of calcium signaling, redox, chromatin remodeling and circadian clock network changes that, together, may account for the strikingly pleiotropic developmental phenotypes and the plant’s capacity for trans-generational stress memory. The authors suggest that sensory plastids participate in sensing environmental stress, integrating this sensory function with epigenetic and gene expression circuitry to condition heritable stress memory.
Plastids differentiate into subtypes. For example, the transition of proplastids to chloroplasts defines the photosynthetic tissues within a plant, whereas amyloplasts are formed in nonphotosynthetic tissues. Recently, evidence has accumulated for a novel class of plastids that is specialized for stress sensing and acclimation. These “sensory plastids”, some of which occur in the epidermis, are approximately 30% the size of mesophyll chloroplasts, and are associated with shade and osmotic stress responses. Similar plastids within the vascular parenchyma have been associated with ABA biosynthesis, and high light and drought responses. Beltrán et al. (10.1104/pp.18.00804) reveal that the distinctive proteome of sensory plastids in Arabidopsis (Arabidopsis thaliana) derives from the spatiotemporal regulation of nuclear genes encoding plastid-targeted proteins. In particular, MutS HOMOLOG 1 (MSH1), a plant-specific mitochondrial protein is associated with this specialized plastid type. The global stress phenotype that derives from MSH1 RNAi knockdown is characterized by genome-wide DNA methylome repatterning, implying an epigenomic component to the phenotype. Investigation of the translatome within sensory plastid-containing cells, following MSH1 disruption, provides evidence of calcium signaling, redox, chromatin remodeling and circadian clock network changes that, together, may account for the strikingly pleiotropic developmental phenotypes and the plant’s capacity for trans-generational stress memory. The authors suggest that sensory plastids participate in sensing environmental stress, integrating this sensory function with epigenetic and gene expression circuitry to condition heritable stress memory.