Protection Against Reactive Carbonyl Species
Schmitz et al. investigate the physiological significance of the glyoxylase system in Arabidopsis https://doi.org/10.1105/tpc.17.00258
By Jessica Schmitz and Veronica G. Maurino

GLXI;3 mutants do not develop when exposed to methylglyoxal (MGO) or glyoxal (GO) and show reduced root size when grown on sucrose.
BACKGROUND: In every living cell, side reactions of enzymes and spontaneous reactions of metabolites inevitably lead to the formation of toxic by-products. To prevent cellular damage, accumulation of these toxic metabolites needs to be tightly controlled through metabolite repair pathways that detoxify unwanted products quickly and efficiently. One important metabolite repair pathway is the glyoxalase system, which is found in all domains of life. This two-step enzymatic system, composed of multiple isoforms of Glyoxalase I (GLXI) and Glyoxalase II (GLXII), converts highly reactive carbonyl species, such as methylglyoxal and glyoxal, to less harmful metabolites. In plants, methylglyoxal is generated during glycolysis and also during the photosynthetic Calvin cycle. To date, knowledge on the importance of the methylglyoxal detoxification pathway in physiological conditions in plants has been fragmentary.
QUESTIONS: To shed light on the physiological significance of the glyoxalase system, we aimed to identify the important glyoxalase isoforms in vivo, their subcellular localizations, and their transcriptional regulation.
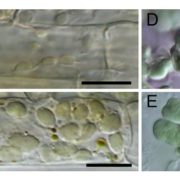
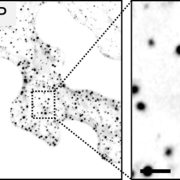
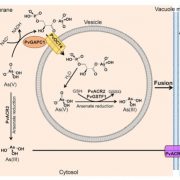
FINDINGS: The glyoxalase system is encoded by three homologs of GLXI and by three homologs of GLXII, from which multiple predicted GLXI and GLXII isoforms can be derived through alternative splicing. We identified the physiologically relevant splice forms using transcriptional data and demonstrated that the resulting isoforms are targeted to different subcellular localizations. We also analyzed their enzymatic properties and demonstrated that efficient methylglyoxal and glyoxal detoxification can be controlled by a switch in metal cofactor usage. Expression analysis indicated that GLXI is transcriptionally regulated by endogenous sugar levels. Analysis of GLXI Arabidopsis loss-of-function mutants revealed that GLXI;3 is crucial for methylglyoxal and glyoxal detoxification especially during germination and seedling establishment. With this information, we propose a cellular model in which methylglyoxal and glyoxal can be detoxified directly at the cellular production site through specific glyoxalase isoforms.
NEXT STEPS: We aim to understand the molecular link between cellular sugar status and physiological situations in which reactive carbonyl species accumulate. Future work will identify the transporters of the glyoxalase system and clarify how the pathway’s enzymatic and transport activities between the involved cellular compartments are synchronized.
Jessica Schmitz, Isabell C Dittmar, Jörn D Brockmann, Marc Schmidt, Meike Hüdig, Alessandro W Rossoni, Veronica G Maurino. (2017). Defense Against Reactive Carbonyl Species Involves at Least Three Subcellular Compartments where Individual Components of the System Respond to Cellular Sugar Status. Plant Cell Published November 2017. https://doi.org/10.1105/tpc.17.00258