Blue Light Perception via Chlorochrome? — Give Us the Greens of Summer
What do photosynthesis, neonatal jaundice, and a next-generation solar cell have in common? All involve tetrapyrroles, complex molecules with four linked pyrrole rings, each ring containing one nitrogen and four carbon atoms. Tetrapyrroles exist in either cyclic or linear form and have a wide variety of biological functions (Tanaka and Tanaka, 2007; Brzezowski, et al., 2015; Larkin, 2016).
Cyclic tetrapyrroles include chlorophyll in a green leaf, vitamin B-12, and heme in a red blood cell. Bilirubin, a breakdown product of heme, is a linear tetrapyrrole (bilin) that often accumulates in newborn infants, causing neonatal jaundice. Bilins, due to their linear conformation, can undergo light-induced cis-trans isomerization at a double bond and thereby act as photosensitive signaling molecules or solar collectors. The Pr/Pfr chromophore of phytochrome is a well known light-sensing bilin, as are the phycobilin-dependent photoreceptors found in cyanobacteria and some nongreen algae.
Now, Wittkopp et al. (10.1105/tpc.17.00149) provide convincing evidence that bilins in Chlamydomonas reinhardtii, which lacks phytochromes, have an unexpected role in synthesizing and sustaining a functional photosynthetic apparatus in the light. Their studies implicate the presence of a bilin-containing photoreceptor they term chlorochrome that regulates chlorophyll accumulation via blue light perception.
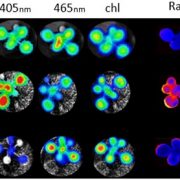
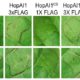
 Previous work showed that the Chlamydomonas heme oxygenase mutant hmox1 is unable to convert heme to the bilin pigment biliverdin (BV) and is also compromised in photoacclimation to high light conditions (Duanmu et al., 2013). Here they show that hmox1 is deficient in light-induced growth, greening, and photosynthesis. These defects were corrected either by introduction of the wild-type HMOX1 cDNA (see figure) or by supplementing cultures with BV, the product of the HMOX1 reaction, as shown in the manuscript.
Previous work showed that the Chlamydomonas heme oxygenase mutant hmox1 is unable to convert heme to the bilin pigment biliverdin (BV) and is also compromised in photoacclimation to high light conditions (Duanmu et al., 2013). Here they show that hmox1 is deficient in light-induced growth, greening, and photosynthesis. These defects were corrected either by introduction of the wild-type HMOX1 cDNA (see figure) or by supplementing cultures with BV, the product of the HMOX1 reaction, as shown in the manuscript.
The hmox1 mutant had reduced photosynthetic O2 evolution in the light and required higher fluence rates to achieve saturation, defects that were more pronounced in higher intensity light. It also had reduced activity of both Photosystems I and II (PSI, PSII) that was rescued by supplementation with BV, with full rescue requiring light. These defects are similar to those of mutants deficient in chlorophyll biosynthesis, except that the hmox1 mutant could be fully rescued by supplementation with biliverdin—a tetrapyrrole that cannot be converted to chlorophyll.
Both dark-grown and light-grown hmox1 cultures were also deficient in several PSI-associated and Light-Harvesting Complex I (LHCI) polypeptides, However, biliverdin could only rescue these defects in the light. Furthermore, the authors showed that both photosystem-antenna supercomplexes (PSII-LHCI and PSI-LHCII) were reduced in hmox1, and that supplementation with BV corrected this in a light-dependent manner.
To determine whether BV rescue in the light depends on light quality, the authors measured levels of PSAD and LHCA1 polypeptides in white, red, and blue light. These studies showed that the BV- and white-light induced restoration of these polypeptides by BV was due specifically to the blue component of the light.
They then conducted a detailed transcriptomic analysis of WT, hmox1 ± BV, and the two complemented lines both to identify differentially expressed genes in light vs. dark and to identify genes whose expression was altered by the absence of HMOX1. Surprisingly, they found that photosynthesis-associated nuclear genes, including genes involved in tetrapyrrole metabolism or photoperception, were unaffected by the hmox1 mutation. Chlorochrome’s blue light-dependency and lack of extensive regulation of nuclear gene expression were in striking contrast with the regulatory properties of phytochromes.
The authors conclude with a compelling model of how bilins and blue light help maintain the integrity of the photosynthetic apparatus in Chlamydomonas. Furthermore, they hypothesize that BV or a subsequent metabolite acts as a chromophore of a blue light-sensing biliprotein (chlorochrome) that activates key steps in chlorophyll biosynthesis. Of particular interest is that neonatal jaundice is treated with blue light, showing that we are not as different from algae as we might think!
REFERENCES
Brzezowski, P., Richter, A.S., and Grimm, B. (2015). Regulation and function of tetrapyrrole biosynthesis in plants and algae. Biochim. Biophys. Acta. 1847: 968-985.
Duanmu, D., et al. (2013). Retrograde bilin signaling enables Chlamydomonas greening and phototrophic survival. Proc. Natl. Acad. Sci. USA 110: 3621-3626.
Larkin, R. M. (2016). Tetrapyrrole signaling in plants. Front. Plant Sci. 7: 1586.
Tanaka, R., and Tanaka, A. (2007). Tetrapyrrole biosynthesis in higher plants. Annu. Rev. Plant. Biol. 58: 321-346.
Wittkopp, T., et al. (2017). Bilin-dependent photoacclimation in Chlamydomonas reinhardtii. The Plant Cell. 10.1105/tpc.17.00149