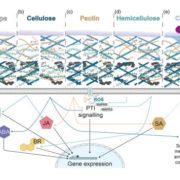
An even pattern of xylan substitution is critical for interaction with cellulose in plant cell walls
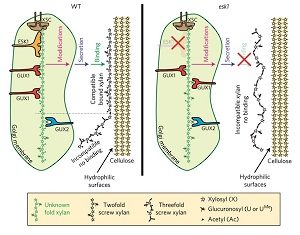 Plant cell wall architecture is a very complex specific design and the interaction between xylan and cellulose is believed to be that way too. Grantham et al. reveal the details of the association between xylan and cellulose using mass spectrometry and NMR in Arabidopsis. ESKIMO1 (ESK1) is a xylan-specific O-acetyltransferase, responsible for adding most of the xylan acetyl groups in a canonical even-pattern distribution which is in phase with 4-O-methylglucuronosyl (U(me)) xylan substitutions. In esk1 mutants, this even-pattern acetylation is lost and plants display a dwarf phenotype with collapsed xylem vessels. Acetylation reduction in esk1 mutants leads to an increase in U(me) and loss of U(me) even-patterning. Through genetic analysis, the authors found that the glucuronosyltransferase GUX1, but not GUX2, is responsible for the even U pattern, likely guided by the acetylation activities of ESK1. Furthermore, the abnormal-patterned xylan substitutions prevented interaction between xylan and cellulose. The authors determined that xylan likely binds the hydrophilic face of cellulose in a manner dependent on this even-patterning of xylan substitutions. Understanding cell wall architecture can be helpful for the processing and application of biomaterials. (Summary by Alecia Biel) Nature Plants. 10.1038/s41477-017-0030-8.
Plant cell wall architecture is a very complex specific design and the interaction between xylan and cellulose is believed to be that way too. Grantham et al. reveal the details of the association between xylan and cellulose using mass spectrometry and NMR in Arabidopsis. ESKIMO1 (ESK1) is a xylan-specific O-acetyltransferase, responsible for adding most of the xylan acetyl groups in a canonical even-pattern distribution which is in phase with 4-O-methylglucuronosyl (U(me)) xylan substitutions. In esk1 mutants, this even-pattern acetylation is lost and plants display a dwarf phenotype with collapsed xylem vessels. Acetylation reduction in esk1 mutants leads to an increase in U(me) and loss of U(me) even-patterning. Through genetic analysis, the authors found that the glucuronosyltransferase GUX1, but not GUX2, is responsible for the even U pattern, likely guided by the acetylation activities of ESK1. Furthermore, the abnormal-patterned xylan substitutions prevented interaction between xylan and cellulose. The authors determined that xylan likely binds the hydrophilic face of cellulose in a manner dependent on this even-patterning of xylan substitutions. Understanding cell wall architecture can be helpful for the processing and application of biomaterials. (Summary by Alecia Biel) Nature Plants. 10.1038/s41477-017-0030-8.