A Key Enzyme in the Biosynthesis of a Plant-Derived anti-HIV Drug
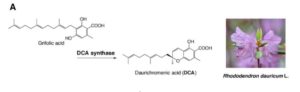 Rhododendron dauricum (Ericaceae), a native of northeastern Asia, produces unique secondary metabolites including daurichromenic acid (DCA). DCA has attracted considerable attention as a medicinal resource because this compound is one of the most effective natural products with anti-HIV properties in cell culture. Thus, chemical synthesis of DCA has been extensively studied over the past few years. Previously, a partial characterization has been made of an oxidocyclase, named DCA synthase, using a crude protein extract from young leaves of R. dauricum. DCA synthase is an enzyme that catalyzes the stereoselective oxidative cyclization of farnesyl moiety of grifolic acid to form DCA. Unlike P-450 type cyclases involved in glyceollin and furanocoumarin biosynthesis, DCA synthase is a soluble protein, and does not need exogenously added cofactors for the reaction. Remarkably, these features are similar to those reported for cannabinoid synthases from Cannabis sativa. Iijima et al. ( isolated the cDNA of the gene encoding DCA synthase, based on homology search against translated R. dauricum young leaf transcriptome, using cannabinoid synthases as queries. Heterologous expression of the recombinant proteins in a Pichia pastoris system provided the evidence that one of the candidate cDNAs is of a gene that encodes an active DCA synthase. Previously, the authors have shown that relevant metabolites as well as DCA synthase activity are predominantly localized to young leaves of R. dauricum that are covered with multicellular trichomes called glandular scales. The authors provide evidence that DCA, a phytotoxic metabolite, is primarily biosynthesized in the glandular scales of young leaves and accumulated extracellularly.
Rhododendron dauricum (Ericaceae), a native of northeastern Asia, produces unique secondary metabolites including daurichromenic acid (DCA). DCA has attracted considerable attention as a medicinal resource because this compound is one of the most effective natural products with anti-HIV properties in cell culture. Thus, chemical synthesis of DCA has been extensively studied over the past few years. Previously, a partial characterization has been made of an oxidocyclase, named DCA synthase, using a crude protein extract from young leaves of R. dauricum. DCA synthase is an enzyme that catalyzes the stereoselective oxidative cyclization of farnesyl moiety of grifolic acid to form DCA. Unlike P-450 type cyclases involved in glyceollin and furanocoumarin biosynthesis, DCA synthase is a soluble protein, and does not need exogenously added cofactors for the reaction. Remarkably, these features are similar to those reported for cannabinoid synthases from Cannabis sativa. Iijima et al. ( isolated the cDNA of the gene encoding DCA synthase, based on homology search against translated R. dauricum young leaf transcriptome, using cannabinoid synthases as queries. Heterologous expression of the recombinant proteins in a Pichia pastoris system provided the evidence that one of the candidate cDNAs is of a gene that encodes an active DCA synthase. Previously, the authors have shown that relevant metabolites as well as DCA synthase activity are predominantly localized to young leaves of R. dauricum that are covered with multicellular trichomes called glandular scales. The authors provide evidence that DCA, a phytotoxic metabolite, is primarily biosynthesized in the glandular scales of young leaves and accumulated extracellularly.






Leave a Reply
Want to join the discussion?Feel free to contribute!