Sulfoxidation of NON-RIPENING Affects Tomato Fruit Ripening
Transcription factors (TFs) can be subject to multiple posttranslational modifications (PTMs) that affect protein stability, subcellular 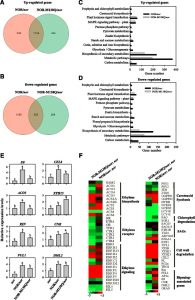 localization, interactions with corepressors and activators, and DNA binding activity of TFs, thereby influencing their regulatory activities on target genes. Recent studies have revealed that redox modification of TFs plays an important role in stress- or disease-related biological processes. These redox modifications are mainly associated with the oxidation of Cys residues in proteins, or the conversion between sulfhydryl and intermolecular/intramolecular disulfide bonds mediated by thioredoxins. Jiang et al. (10.1104/pp.20.00070) now establish that NON-RIPENING (NOR), a master TF that regulates tomato (Solanum lycopersicum) fruit ripening, is a target of the Met sulfoxide reductases A (E4) and B (SlMsrB2) in tomato. Met oxidation in NOR (i.e. sulfoxidation), or mimicking sulfoxidation by mutating Met-138 to Gln, reduces its DNA-binding capacity and transcriptional regulatory activity in vitro. E4 and SlMsrB2 partially repair oxidized NOR and restore its DNA-binding capacity. Transgenic complementation of the nor mutant with NOR partially rescues the ripening defects. However, transformation of nor with NOR-M138Q, which mimicks the effects of Met sulfoxidation, inhibits restoration of the fruit ripening phenotype, and this is associated with the decreased DNA-binding and transcriptional activation of a number of ripening-related genes. Taken together, these observations reveal a PTM mechanism by which MsrB2-mediated redox modification of NOR regulates the expression of ripening-related genes, thereby influencing tomato fruit ripening.
localization, interactions with corepressors and activators, and DNA binding activity of TFs, thereby influencing their regulatory activities on target genes. Recent studies have revealed that redox modification of TFs plays an important role in stress- or disease-related biological processes. These redox modifications are mainly associated with the oxidation of Cys residues in proteins, or the conversion between sulfhydryl and intermolecular/intramolecular disulfide bonds mediated by thioredoxins. Jiang et al. (10.1104/pp.20.00070) now establish that NON-RIPENING (NOR), a master TF that regulates tomato (Solanum lycopersicum) fruit ripening, is a target of the Met sulfoxide reductases A (E4) and B (SlMsrB2) in tomato. Met oxidation in NOR (i.e. sulfoxidation), or mimicking sulfoxidation by mutating Met-138 to Gln, reduces its DNA-binding capacity and transcriptional regulatory activity in vitro. E4 and SlMsrB2 partially repair oxidized NOR and restore its DNA-binding capacity. Transgenic complementation of the nor mutant with NOR partially rescues the ripening defects. However, transformation of nor with NOR-M138Q, which mimicks the effects of Met sulfoxidation, inhibits restoration of the fruit ripening phenotype, and this is associated with the decreased DNA-binding and transcriptional activation of a number of ripening-related genes. Taken together, these observations reveal a PTM mechanism by which MsrB2-mediated redox modification of NOR regulates the expression of ripening-related genes, thereby influencing tomato fruit ripening.



