Genome-Wide Analysis of Temperature-Induced Fatty Acid Desaturation
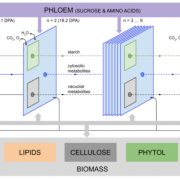
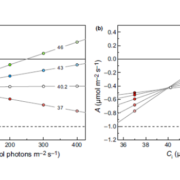
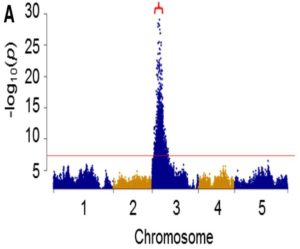 Biogeographic studies suggest that the degree of fatty acid unsaturation in oilseeds has played a major role in adapting plants to temperature. The enzymes that are primarily responsible for producing polyunsaturated fatty acids in seed oils are the microsomal ω-6 and ω-3 fatty acid desaturases FAD2 and FAD3, which act in sequence to convert oleic acid (18:1) to linoleic acid (18:2) and then to α-linolenic acid (18:3), while the fatty acid is esterified to the sn-2 position of phosphatidylcholine. Temperature can influence the degree of microsomal fatty acid desaturation indirectly through its effects on substrate availability. However, there also is evidence that FAD2 and FAD3 are regulated directly by temperature. Whether this regulation occurs at the level of gene expression appears to vary depending upon the species, tissue, and gene in question. Many previous studies have exploited genetic approaches to investigate the function of fatty acid desaturation in plant temperature adaptation. However, no studies appear to have used genetic variation in plants to study the underlying regulatory mechanisms. Menard et al. (Plant Physiol. 173: 1594–1605) have characterized the response of Arabidopsis seed lipids to variation in ambient temperature and found that warm temperatures inhibit both ω-6 and ω-3 desaturation in phosphatidylcholine. Analyses of 19 parental accessions showed that significant natural variation exists in the temperature responsiveness of ω-6 desaturation. A combination of quantitative trait locus analysis and genome-wide association studies suggests that ω-6 desaturation is largely controlled by cis-acting sequence variants in the FAD2 5′ untranslated region intron that determines the expression level of the gene. However, the temperature responsiveness of ω-6 desaturation is controlled by a separate quantitative trait locus on chromosome 2.
Biogeographic studies suggest that the degree of fatty acid unsaturation in oilseeds has played a major role in adapting plants to temperature. The enzymes that are primarily responsible for producing polyunsaturated fatty acids in seed oils are the microsomal ω-6 and ω-3 fatty acid desaturases FAD2 and FAD3, which act in sequence to convert oleic acid (18:1) to linoleic acid (18:2) and then to α-linolenic acid (18:3), while the fatty acid is esterified to the sn-2 position of phosphatidylcholine. Temperature can influence the degree of microsomal fatty acid desaturation indirectly through its effects on substrate availability. However, there also is evidence that FAD2 and FAD3 are regulated directly by temperature. Whether this regulation occurs at the level of gene expression appears to vary depending upon the species, tissue, and gene in question. Many previous studies have exploited genetic approaches to investigate the function of fatty acid desaturation in plant temperature adaptation. However, no studies appear to have used genetic variation in plants to study the underlying regulatory mechanisms. Menard et al. (Plant Physiol. 173: 1594–1605) have characterized the response of Arabidopsis seed lipids to variation in ambient temperature and found that warm temperatures inhibit both ω-6 and ω-3 desaturation in phosphatidylcholine. Analyses of 19 parental accessions showed that significant natural variation exists in the temperature responsiveness of ω-6 desaturation. A combination of quantitative trait locus analysis and genome-wide association studies suggests that ω-6 desaturation is largely controlled by cis-acting sequence variants in the FAD2 5′ untranslated region intron that determines the expression level of the gene. However, the temperature responsiveness of ω-6 desaturation is controlled by a separate quantitative trait locus on chromosome 2.





Leave a Reply
Want to join the discussion?Feel free to contribute!