Coordinating Cell Walls and Cell Growth: A Role for LRX Extensin Chimeras
To regulate the addition of cell wall components as cells expand and to control changes in cell wall composition as cells differentiate, there must be a feedback system that senses the state of the cell wall and transmits this information. The mechanisms underlying cell wall-intracellular signaling remain enigmatic despite the fundamental importance of such a system for normal growth. Cell wall proteins must play a role in this process. The most abundant cell wall proteins are extensins, but their relatively simple structure and the fact that they are covalently cross-linked within the cell wall suggests that they have a role in stabilization rather than signaling.
The LRX (for Leu-rich extensin chimera) proteins contain a C-terminal extensin domain and are tightly bound to cell walls, but their N-terminal Leu-rich repeat (LRR) domain presumably recognizes a ligand, and this interaction may participate in a cell wall signaling system (Rubinstein et al., 1995a, 1995b; Ringli, 2010). Consistent with this idea, genetic and biochemical studies have linked LRX proteins to cell growth and cell wall composition in growing root hairs and in overall plant growth (Baumberger et al., 2001; Draeger, et al., 2015).
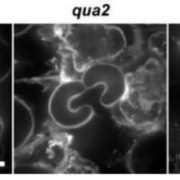
In this issue of Plant Physiology, Wang et al. (2018) and Fabrice et al. (2018) examine the role of LRX proteins in pollen tube growth. Pollen tubes growing in vitro constitute a powerful model system for probing cell wall signaling due to the ease of manipulating growth conditions and imaging pollen tube growth in real time. Both groups carefully analyzed triple and quadruple mutations in the four pollen-expressed LRX genes in Arabidopsis, LRX8, LRX9, LRX10, and LRX11. Their results clearly indicate that LRX proteins are required for pollen tube germination and growth, a conclusion also supported in a brief paper by Sede et al. (2018). Abnormalities in mutant pollen tube growth in vitro included pollen tube bulging, branching, and bursting, phenotypes congruent with defects in cell wall structure. Fabrice et al. delved deeper into mutant phenotypes, documenting abnormalities in pollen and pollen tube cell walls at the ultrastructural level, as well as abnormal vesicle dynamics in growing tubes, and a Ca2+ spike just prior to pollen tube bursting.
Fabrice et al. further presented evidence supporting the possibility that LRX proteins may link the cell wall and the cell membrane by showing that the extensin domain is required for cell wall localization of LRX proteins, but when extensin domain is removed, the LRR domain preferentially associates with a membrane fraction in cell lysates. Since the LRR domain itself is unlikely to bind directly to membranes, this result suggests that LRX proteins may act in a complex that includes the cell membrane proteins. If such a complex occurs, it is possible that other members may include proteins identified through work presented in two recent Science papers. Mecchia et al. (2017) show that pollen-expressed RALF (for rapid alkalinization factor) peptides AtRALF4/19 are required to prevent pollen tube bursting. Pollen RALF peptides were originally identified as potential LRX binding partners using a tomato (Solanum lycopersicum) pollen LRX protein as bait in a yeast two-hybrid screen (Covey et al., 2010), and Mecchia et al. (2017) confirm the physical interaction of AtRALF4/19 peptides with LRX8. Ge et al. (2017) show that these same AtRALF4/19 peptides also interact with pollen-expressed transmembrane CrRLK1L family receptors ANX1/2 and BUPS1/2, which have carbohydrate-binding malectin ectodomains.
These converging results support the notion of a multicomponent cell wall signaling complex consisting of mobile extracellular RALF peptides, cell wall-bound LRX proteins, and carbohydrate-binding transmembrane CrRLK1L family receptors. Exactly how such a complex senses structural changes in cell walls will be the next big mystery to solve.
REFERENCES
Baumberger N, Ringli C, Keller B (2001) The chimeric leucine-rich repeat/extensin cell wall protein LRX1 is required for root hair morphogenesis in Arabidopsis thaliana. Genes Dev 15: 1128–1139
Covey PA, Subbaiah CC, Parsons RL, Pearce G, Lay FT, Anderson MA, Ryan CA, Bedinger PA (2010) A pollen-specific RALF from tomato that regulates pollen tube elongation. Plant Physiol 153: 703–715
Draeger C, Ndinyanka Fabrice T, Gineau E, Mouille G, Kuhn BM, Moller I, Abdou M-T, Frey B, Pauly M, Bacic A, et al. (2015) Arabidopsis leucine-rich repeat extensin (LRX) proteins modify cell wall composition and influence plant growth. BMC Plant Biol 15: 155
Fabrice TN, Vogler H, Draeger C, Munglani G, Gupta S, Herger AG, Knox P, Grossniklaus U, Ringl C (2018) LRX proteins play a crucial role in pollen grain and pollen tube cell wall development. Plant Physiol 176: 1981–1992
Ge Z, Bergonci T, Zhao Y, Zou Y, Du S, Liu MC, Luo X, Ruan H, García-Valencia LE, Zhong S, et al. (2017) Arabidopsis pollen tube integrity and sperm release are regulated by RALF-mediated signaling. Science 358: 1596–1600
Mecchia MA, Santos-Fernandez G, Duss NN, Somoza SC, Boisson-Dernier A, Gagliardini V, Martínez-Bernardini A, Fabrice TN, Ringli C, Muschietti JP, et al. (2017) RALF4/19 peptides interact with LRX proteins to control pollen tube growth in Arabidopsis. Science 358: 1600–1603
Rubinstein AL, Broadwater AH, Lowrey KB, Bedinger PA (1995a) Pex1, a pollen-specific gene with an extensin-like domain. Proc Natl Acad Sci USA 92: 3086–3090
Rubinstein AL, Marquez J, Suarez-Cervera M, Bedinger PA (1995b) Extensin-like glycoproteins in the maize pollen tube wall. Plant Cell 7: 2211–2225
Ringli C (2010) The hydroxyproline-rich glycoprotein domain of the Arabidopsis LRX1 requires Tyr for function but not for insolubilization in the cell wall. Plant J 63: 662–669
Sede AR, Borassi C, Wengier DL, Mecchia MA, Estevez JM, Muschietti JP (2018) Arabidopsis pollen extensins LRX are required for cell wall integrity during pollen tube growth. FEBS Lett 592: 233–243
Wang X, Wang K, Yin G, Liu X, Liu M, Cao N, Duan Y, Gao H, Wang W, Ge W, et al. (2018) Pollen-expressed leucine-rich repeat extensins are essential for pollen germination and growth. Plant Physiol 176: 1993–2006