Concentration-dependent transcriptional switching through a collective action of cis-elements (Sci. Adv.)
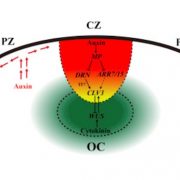
 One of the well-known molecular pathways that regulate plant development is the one formed by the homeobox transcription factor WUSCHEL (WUS) and the secreted peptide CLAVATA 3 (CLV3). In the shoot apical meristem (SAM), WUS expression is constrained to the organizing center where it maintains the stem cell population. WUS protein migrates to adjacent cells creating a concentration gradient that activates CLV3, its own negative regulator. But, at the same time, high WUS concentration leads to protein dimerization and CLV3 repression. Here, Rodriguez et al. combine biochemical analyses together with exhaustive computational modeling to answer the question of how WUS levels guide this balance between CLV3 transcriptional activation and repression. WUS binds to a module of five cis-elements in the CLV3 promoter. The authors demonstrate that the individual affinity of each site as well as the number of elements define CLV3 expression. Furthermore, the spacing between these cis-elements is also critical since it determines the cooperativity of WUS binding. Higher distance increases the affinity for WUS monomers, which promotes CLV3 activation, thus revealing a new cofactor-independent mechanism of regulation of transcription factor binding. Overall, their models reveal the particular conditions required to recreate CLV3 spatial expression specificity. This knowledge can be used in the future to precisely engineer the stem cell dynamics in the SAM, thereby modulating plant growth. (Summary by Daniel Alique, @daniel_alique) Science Advances 10.1126/sciadv.abo6157.
One of the well-known molecular pathways that regulate plant development is the one formed by the homeobox transcription factor WUSCHEL (WUS) and the secreted peptide CLAVATA 3 (CLV3). In the shoot apical meristem (SAM), WUS expression is constrained to the organizing center where it maintains the stem cell population. WUS protein migrates to adjacent cells creating a concentration gradient that activates CLV3, its own negative regulator. But, at the same time, high WUS concentration leads to protein dimerization and CLV3 repression. Here, Rodriguez et al. combine biochemical analyses together with exhaustive computational modeling to answer the question of how WUS levels guide this balance between CLV3 transcriptional activation and repression. WUS binds to a module of five cis-elements in the CLV3 promoter. The authors demonstrate that the individual affinity of each site as well as the number of elements define CLV3 expression. Furthermore, the spacing between these cis-elements is also critical since it determines the cooperativity of WUS binding. Higher distance increases the affinity for WUS monomers, which promotes CLV3 activation, thus revealing a new cofactor-independent mechanism of regulation of transcription factor binding. Overall, their models reveal the particular conditions required to recreate CLV3 spatial expression specificity. This knowledge can be used in the future to precisely engineer the stem cell dynamics in the SAM, thereby modulating plant growth. (Summary by Daniel Alique, @daniel_alique) Science Advances 10.1126/sciadv.abo6157.