Which Factors Control Starch Granule Initiation?
Storage and remobilization of sugar molecules play important roles for the growth and survival of living organisms. Besides a few exceptions, animals store carbohydrates in the form of soluble glycogen while green plants and algae bank glucans as insoluble starch. Starch forms the basis of human nutrition and constitutes a renewable raw material for industry thus highlighting its importance for humankind.
Starch is a non-structural polymer composed of linked glucose units forming inert granules within the chloroplast. In leaves, transitory starch is synthesized during the day from photosynthetic assimilates and subsequently degraded to provide energy for continuous growth at night (reviewed in Pfister and Zeeman, 2016). The enzymes controlling starch synthesis and degradation have been extensively studied and recent attention has also been given to factors controlling starch granule initiation. Notably, plants lacking PROTEIN TARGETING TO STARCH 2 (PTST2) or its interaction partners MYOSIN-RESEMBLING CHLOROPLAST PROTEIN (MRC) and MAR BINDING FILAMENT-LIKE PROTEIN 1 (MFP1) have a reduced number of starch granules per chloroplast (Seung et al., 2017, 2018; Vandromme et al., 2019). In this issue of The Plant Cell, Abt et al., (2020) characterize the function of STARCH SYNTHASE 5 (SS5) in starch metabolism and show that this inactive enzyme plays an important role within the nexus of factors regulating the number of starch granules formed in chloroplasts.
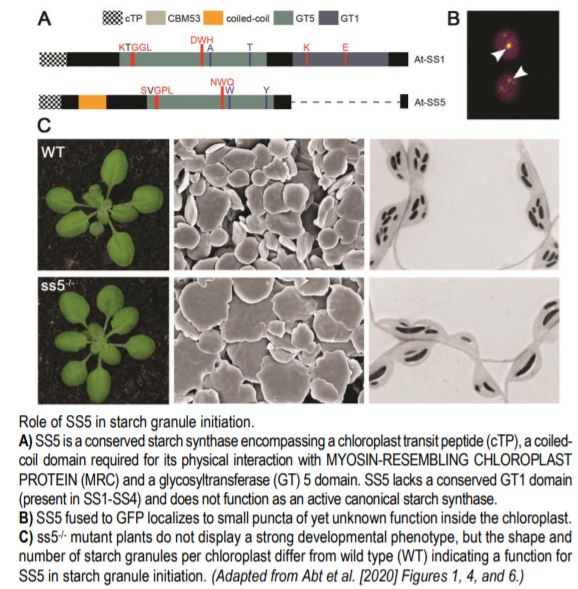 The plant model organism Arabidopsis thaliana contains six starch synthases. Five of them participate in elongating glucan chains to make amylopectin and amylose, the two polysaccharide types found in starch. Abt and colleague observed that the sixth starch synthase, SS5, was found throughout the plant kingdom but lacked a conserved glycosyltransferase (GT) 1 domain (see Figure). Structural modeling indicated that SS5 has a surface carbohydrate binding site conserved with SS4. Consistently, recombinant SS5 was able to bind maize starch but not to actively transfer ADP-glucose onto it. Furthermore, SS5 was not able to produce glucans when expressed in yeast confirming that SS5 was not an active glucosyltransferase.
The plant model organism Arabidopsis thaliana contains six starch synthases. Five of them participate in elongating glucan chains to make amylopectin and amylose, the two polysaccharide types found in starch. Abt and colleague observed that the sixth starch synthase, SS5, was found throughout the plant kingdom but lacked a conserved glycosyltransferase (GT) 1 domain (see Figure). Structural modeling indicated that SS5 has a surface carbohydrate binding site conserved with SS4. Consistently, recombinant SS5 was able to bind maize starch but not to actively transfer ADP-glucose onto it. Furthermore, SS5 was not able to produce glucans when expressed in yeast confirming that SS5 was not an active glucosyltransferase.
Plants lacking SS5 did not display a strong visible phenotype (see Figure) and overall starch levels remained similar to wild-type, except at the end of the night where ss5-/- had slightly more starch. Scanning electron micrographs of purified starch granules and light micrographs of embedded tissue sections showed that ss5-deficient plants had fewer but larger starch granules (see Figure). Together, this data suggested that SS5 was not primarily involved in starch synthesis or degradation but rather in starch granule initiation.
Immunoprecipitation followed by mass spectrometry of tagged SS5 revealed an interaction with SS4 and MRC. Co-immunoprecipitation experiments validated the interaction between SS5 and MRC and showed that the coiled-coil domain of SS5 was required for this interaction. In vivo localization experiments of fluorescent transcriptional fusions revealed that SS5 was localized in the chloroplast, consistent with the present of an N-terminal chloroplast transit peptide (see Figure). Interestingly, the authors found that SS5 and MRC co-localized in small puncta suggesting the existence of a possible structural “platform” regulating starch granule homeostasis.
To understand the functional relationship between SS5 and known granule-initiation factors, the authors created several high-order mutants with ss5-/- and quantified the number and size of starch granules per chloroplast. Interestingly, the additional loss of SS5 did not enhance the phenotype of mrc-/- indicating that SS5 and MRC function in the same pathway. A similar epistatic relationship was observed between SS5 and MFP1. In contrast, the loss of SS4 or PTST2 in addition to SS5 increased the phenotype of the single ss5-/- mutant suggesting that SS5, SS4 and PTST2 can act independently in starch granule initiation.
In summary, Abt and colleagues coined SS5 as a new player in the network of factors determining starch granule initiation. Future investigations will tackle the complex relationships between all these factors and also likely shed light on the exact nature of SS5-MRC containing puncta, whose function remains enigmatic.
Sylvain Bischof
Assistant Features Editor
Department of Plant and Microbial Biology
University of Zürich, Switzerland
ORCHID ID: 0000-0003-2910-5132
REFERENCES
Pfister, B. and Zeeman, S. C. (2016). Formation of starch in plant cells. Cell. Mol. Life Sci. 73: 2781-2807.
Seung, D. et al (2017) Homologs of PROTEIN TARGETING TO STARCH Control Starch Granule Initiation in Arabidopsis Leaves. Plant Cell 29: 1657-1677.
Seung, D. et al. (2018) Two Plastidial Coiled- Coil Proteins Are Essential for Normal Starch Granule Initiation in Arabidopsis. Plant Cell 30: 1523-1542.
Vandromme, C. et al. (2019) PII1: a protein involved in starch initiation that determines granule number and size in Arabidopsis chloroplast. New Phytol. 221: 356-370.
Abt, M.R. et al. (2020) STARCH SYNTHASE 5, a Noncanonical Starch Synthase-Like Protein Is 4 Involved in Starch Granule Initiation in Arabidopsis. Plant Cell DOI https://doi.org/10.1105/tpc.19.00946.