TOR promotes guard cell starch degradation and stomatal opening
Chao Han and Ming-yi Bai
The Key Laboratory of Plant Development and Environmental Adaptation Biology, Ministry of Education, School of Life Sciences, Shandong University, Qingdao, 266237, China.
Background: Starch is widely present in guard cells of different plant species. Rapid starch degradation in guard cells is crucial for stomatal opening upon light exposure in plants. Guard cell specific Target of rapamycin (TOR) is an evolutionarily conserved kinase that modulates nutrient status and energy signaling to regulate growth and development in all eukaryotes. In Arabidopsis, TOR inactivation arrests plant growth and leads to starch and triacylglycerol (TAG) accumulation in whole seedlings. However, it is unclear whether sugar and TOR regulate starch metabolism in guard cells and stomatal movement.
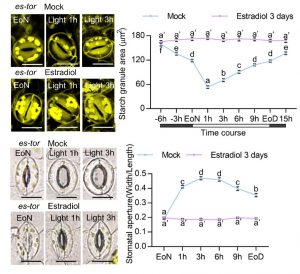 Question: Does sugar-activated TOR regulate guard cell starch degradation and stomatal opening? We tested this by measuring the starch granules and stomatal apertures of plants in which TOR had been inactivated by estradiol-induced RNA interference or AZD8055 treatment.
Question: Does sugar-activated TOR regulate guard cell starch degradation and stomatal opening? We tested this by measuring the starch granules and stomatal apertures of plants in which TOR had been inactivated by estradiol-induced RNA interference or AZD8055 treatment.
Findings: TOR inactivation or a lack of carbon caused by a short photoperiod or low light intensity resulted in guard cell starch accumulation and impaired stomatal opening upon light exposure. Sugar-activated TOR kinase induces BAM1 expression by stabilizing BRASSINAZOLE-RESISTANT1 (BZR1), the key transcriptional factor in brassinosteroid signaling, and increases BAM1 protein stability via inducing the phosphorylation of BAM1 at Ser-31, thereby promoting guard cell starch degradation and stomatal opening.
Next steps: Based on these findings, we will carry out research to identify the key kinase or phosphatase responsible for the phosphorylation levels of BAM1, and perform some experiments to examine how to use sugar to improve photosynthetic efficiency.
Chao Han, Wenbo Hua, Jinge Li, Yan Qiao, Lianmei Yao, Wei Hao, Ruizhi Li, Min Fan, Geert De Jaeger, Wenqiang Yang and Ming-Yi Bai. (2022). TOR promotes guard cell starch degradation by regulating the activity of β-AMYLASE1 in Arabidopsis. https://doi.org/10.1093/plcell/koab307




