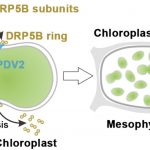
The Smell of a Peach or Not
Fruit aromas attract seed-dispersal agents in nature and are critical determinants of human fruit choice. In the process of constructing a peach (Prunus persica) core germplasm collection, Peng et al. (10.1104/pp.19.00964) analyzed the characteristics of peach cultivars in 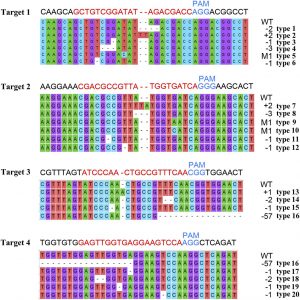 the National Fruit Germplasm Repository (Nanjing, China) and found that, since the 1980s, some varieties have lost their peach-like aroma. Previous analyses of peach fruit volatiles identified more than 100 compounds and confirmed g-decalactone as the most important component contributing to peach aroma. Comparison of the transcriptomes and metabolomes of a high-aroma peach cultivar, Fenghuayulu, and a low-aroma cultivar, Achutao, suggested that amino acid substitutions in ALCOHOL ACYLTRANSFERASE (PpAAT1) are responsible for the undetectable levels of g-decalactone in cv. Achutao fruit. In vitro enzyme assays on PpAAT1 heterologously expressed and purified from Escherichia coli and in vivo assays using transient PpAAT1 expression in Nicotiana benthamiana or the oleaginous yeast Yarrowia lipolytica indicated that PpAAT1 from high-aroma cultivars was more efficient than PpAAT1 from low-aroma cultivars in catalyzing the conversion of 4-hydroxydecanoyl-coenzyme A into g-decalactone. Examination of loss-of-function mutations of PpAAT1 generated by CRISPR/Cas9 in cv. Fenghuayulu showed that fruits with PpAAT1 mutations had significantly lower g-decalactone contents. Expression of the version of PpAAT1 from cv. Fenghuayulu in cv. Achutao restored g-decalactone levels to those measured in ‘Fenghuayulu’, confirming the specific contribution of PpAAT1 to the formation of this key aroma compound.
the National Fruit Germplasm Repository (Nanjing, China) and found that, since the 1980s, some varieties have lost their peach-like aroma. Previous analyses of peach fruit volatiles identified more than 100 compounds and confirmed g-decalactone as the most important component contributing to peach aroma. Comparison of the transcriptomes and metabolomes of a high-aroma peach cultivar, Fenghuayulu, and a low-aroma cultivar, Achutao, suggested that amino acid substitutions in ALCOHOL ACYLTRANSFERASE (PpAAT1) are responsible for the undetectable levels of g-decalactone in cv. Achutao fruit. In vitro enzyme assays on PpAAT1 heterologously expressed and purified from Escherichia coli and in vivo assays using transient PpAAT1 expression in Nicotiana benthamiana or the oleaginous yeast Yarrowia lipolytica indicated that PpAAT1 from high-aroma cultivars was more efficient than PpAAT1 from low-aroma cultivars in catalyzing the conversion of 4-hydroxydecanoyl-coenzyme A into g-decalactone. Examination of loss-of-function mutations of PpAAT1 generated by CRISPR/Cas9 in cv. Fenghuayulu showed that fruits with PpAAT1 mutations had significantly lower g-decalactone contents. Expression of the version of PpAAT1 from cv. Fenghuayulu in cv. Achutao restored g-decalactone levels to those measured in ‘Fenghuayulu’, confirming the specific contribution of PpAAT1 to the formation of this key aroma compound.