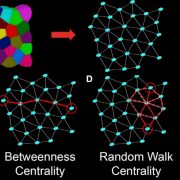
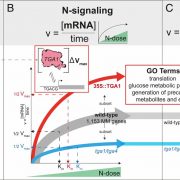
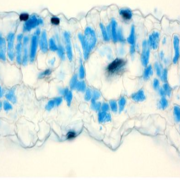
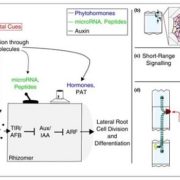
Systems Biology of Deetiolation
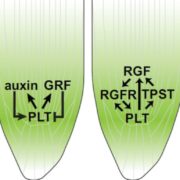
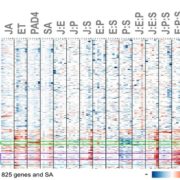
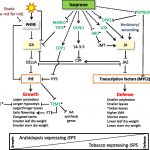
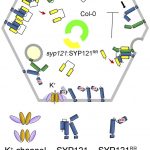
Upon exposure to light, many leaf and stem cells acquire photosynthetic competence by converting pale etioplasts into green chloroplasts. Deetiolation involves the concerted and synchronized activity of a highly complex biogenesis program. Thylakoid membranes must develop from disassembling prolamellar bodies and prothylakoids and from newly synthesized lipids. Moreover, large protein complexes containing dozens of protein subunits and hundreds of pigments and cofactors must be inserted into the budding membrane in strictly defined 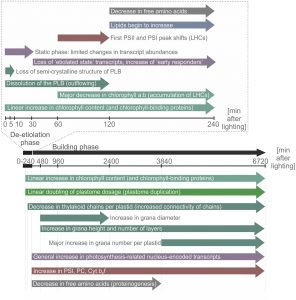 stoichiometric ratios. The protein complexes involved consist of polypeptides originating from two evolutionarily distinct compartments, the nucleus and the plastid, which must be expressed, processed, targeted, and inserted into the membrane in a highly coordinated manner. These processes are dependent upon and controlled by a wide range of assembly chaperones and other biogenesis factors, which are not or only poorly understood. In spite of the complexity of thylakoid biogenesis, the etioplast-to-chloroplast transition can occur astoundingly rapidly. Armarego-Marriott et al. (10.1104/pp.18.01432) have developed a system to study both the deetiolation process and the process of photosynthetic maturation in leaves of tobacco at high temporal resolution. Targeted and nontargeted approaches were undertaken to define the dynamic changes in the transcriptomes of the nucleus and the plastid. In addition, the accumulation kinetics of pigments, lipids, soluble metabolites, and photosynthetic proteins and their activities were determined and correlated with the physical changes in membrane ultrastructure. This work provides a comprehensive systems-level description of thylakoid development and the etioplast-to-chloroplast differentiation process and also reveals candidate genes involved in chloroplast biogenesis and the acquisition of photosynthetic competence.
stoichiometric ratios. The protein complexes involved consist of polypeptides originating from two evolutionarily distinct compartments, the nucleus and the plastid, which must be expressed, processed, targeted, and inserted into the membrane in a highly coordinated manner. These processes are dependent upon and controlled by a wide range of assembly chaperones and other biogenesis factors, which are not or only poorly understood. In spite of the complexity of thylakoid biogenesis, the etioplast-to-chloroplast transition can occur astoundingly rapidly. Armarego-Marriott et al. (10.1104/pp.18.01432) have developed a system to study both the deetiolation process and the process of photosynthetic maturation in leaves of tobacco at high temporal resolution. Targeted and nontargeted approaches were undertaken to define the dynamic changes in the transcriptomes of the nucleus and the plastid. In addition, the accumulation kinetics of pigments, lipids, soluble metabolites, and photosynthetic proteins and their activities were determined and correlated with the physical changes in membrane ultrastructure. This work provides a comprehensive systems-level description of thylakoid development and the etioplast-to-chloroplast differentiation process and also reveals candidate genes involved in chloroplast biogenesis and the acquisition of photosynthetic competence.