Low Xylem Vulnerability in Oaks
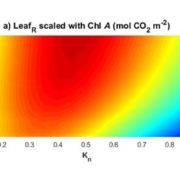
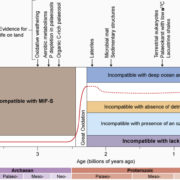
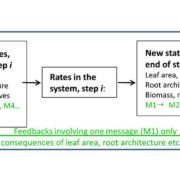
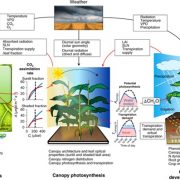
 Under conditions of drought stress, the continuous column of water in the plant xylem experiences increasing tension caused by declining water potential at the sites of evaporation. Eventually, air is drawn into the water transport system, forming embolisms in the xylem conduits. Although plants have developed several mechanisms to restore vessel functionality by refilling embolized vessels, those that have experienced a loss of hydraulic conductance due to embolism formation within the xylem do not always recover full hydraulic functionality following rehydration and often suffer reduced gas exchange capacity as a result. The capacity of plants to resist embolism formation in the xylem is hypothesized to be a major component of plant drought tolerance and survival. An interesting question concerns the capacity of ring-porous species such as oak (Quercus spp.) to resist embolism: these species tend to have long, wide-bored vessels that one might suppose are more likely to form embolisms. In this issue, Skelton et al. (10.1104/pp.18.00103) investigate vulnerability to embolism in Quercus. Their main objectives were to examine how the capacity to withstand embolism within leaves and stems varies across Quercus species and whether vulnerability to embolism in leaves and stems is a critical component of drought tolerance in this ecologically important genus. They selected eight deciduous and evergreen Quercus species with varying climatic niches from the Mediterranean-type climate region of California (USA) and quantified leaf and stem xylem vulnerability to embolism using newly developed optical techniques. Embolism occurred earlier in leaves than in stems in only one out of eight sample species, and plants always lost turgor before experiencing stem embolism. In toto, their results suggest that North American Quercus species are more resistant to embolism than previously thought and support the hypothesis that avoiding stem embolism is a critical component of drought tolerance in woody trees.
Under conditions of drought stress, the continuous column of water in the plant xylem experiences increasing tension caused by declining water potential at the sites of evaporation. Eventually, air is drawn into the water transport system, forming embolisms in the xylem conduits. Although plants have developed several mechanisms to restore vessel functionality by refilling embolized vessels, those that have experienced a loss of hydraulic conductance due to embolism formation within the xylem do not always recover full hydraulic functionality following rehydration and often suffer reduced gas exchange capacity as a result. The capacity of plants to resist embolism formation in the xylem is hypothesized to be a major component of plant drought tolerance and survival. An interesting question concerns the capacity of ring-porous species such as oak (Quercus spp.) to resist embolism: these species tend to have long, wide-bored vessels that one might suppose are more likely to form embolisms. In this issue, Skelton et al. (10.1104/pp.18.00103) investigate vulnerability to embolism in Quercus. Their main objectives were to examine how the capacity to withstand embolism within leaves and stems varies across Quercus species and whether vulnerability to embolism in leaves and stems is a critical component of drought tolerance in this ecologically important genus. They selected eight deciduous and evergreen Quercus species with varying climatic niches from the Mediterranean-type climate region of California (USA) and quantified leaf and stem xylem vulnerability to embolism using newly developed optical techniques. Embolism occurred earlier in leaves than in stems in only one out of eight sample species, and plants always lost turgor before experiencing stem embolism. In toto, their results suggest that North American Quercus species are more resistant to embolism than previously thought and support the hypothesis that avoiding stem embolism is a critical component of drought tolerance in woody trees.