Glyphosate Resistance in Barnyard Grass
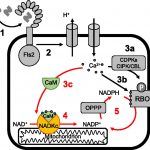
Glyphosate is the world’s most commonly used herbicide, owing to its high efficacy, broad spectrum, and systemic mode of action. Most plant species cannot significantly metabolize glyphosate, which is a major factor contributing to its lethality in plants. However, the 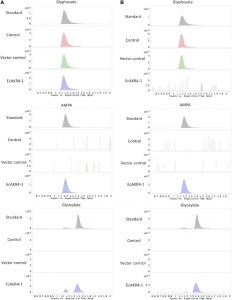 widespread adoption of glyphosate-tolerant transgenic crops from 1996 onwards imposed very high glyphosate selection pressure in agricultural systems, resulting in the extensive evolution of glyphosate resistance in weeds. Given the widespread occurrence and importance of glyphosate-resistant weed evolution, the biochemical and molecular basis of mechanisms endowing glyphosate resistance is under intensive study. It has been determined that specific mutations in the target enzyme of glyphosate, 5-enolpyruvylshikimate3-P synthase (EPSPS), often endow glyphosate resistance to plants. An Australian population of Echinochloa colona (awnless barnyard grass), however, has evolved resistance to glyphosate by a mechanism that does not involve EPSPS or reduced glyphosate uptake or translocation at the tissue level. By isolating and comparing glyphosate-resistant (GR) and -susceptible (GS) E. colona lines, Pan et al. (10.1104/pp.19.00979), using RNA sequencing (RNA-seq), have identified a novel AKR (Aldoketo
widespread adoption of glyphosate-tolerant transgenic crops from 1996 onwards imposed very high glyphosate selection pressure in agricultural systems, resulting in the extensive evolution of glyphosate resistance in weeds. Given the widespread occurrence and importance of glyphosate-resistant weed evolution, the biochemical and molecular basis of mechanisms endowing glyphosate resistance is under intensive study. It has been determined that specific mutations in the target enzyme of glyphosate, 5-enolpyruvylshikimate3-P synthase (EPSPS), often endow glyphosate resistance to plants. An Australian population of Echinochloa colona (awnless barnyard grass), however, has evolved resistance to glyphosate by a mechanism that does not involve EPSPS or reduced glyphosate uptake or translocation at the tissue level. By isolating and comparing glyphosate-resistant (GR) and -susceptible (GS) E. colona lines, Pan et al. (10.1104/pp.19.00979), using RNA sequencing (RNA-seq), have identified a novel AKR (Aldoketo
Reductase) gene (designated as EcAKR4-1) in their GR E. colona population. Rice (Oryza sativa) calli and seedlings overexpressing EcAKR4-1 and displaying increased AKR activity were resistant to glyphosate. EcAKR4-1 expressed in Escherichia coli was found to metabolize glyphosate to produce aminomethylphosphonic acid (AMPA) and glyoxylate. Consistent with these results, GR E. colona plants exhibited enhanced capacity for detoxifying glyphosate into AMPA and glyoxylate. This study provides direct experimental evidence of the evolution of a plant AKR that metabolizes glyphosate and thereby confers glyphosate resistance.