Exploring plant chemistry with a red cabbage pH indicator: Rainbows of color via plant chemistry
A hands-on activity posted by Lucas Busta @PlantsRChemists
In this activity, participants will work with test tubes of red cabbage juice to determine the pH of various other (plant-based) liquids and solutions. It’s a fun activity for all ages – even if participants are too young to understand acid/base chemistry, they really enjoy the color changes and working with test tubes!

Equipment
- 1 red cabbage
- 1 little bottle of lemon juice (optional: and one lemon)
- 1 bottle of watered-down bleach*
- 1 bottle of green tea
- 1 bottle of coffee
- … bottles of other (ideally transparent, plant-based) things with a pH other than 7 (cell culture supernatants, vinegar, etc)
- Paper towels
- 5-6 weigh boats
- solo cups
- 2-4 test tube racks
- 24 test tubes (that fit in the racks)
- 6-8 Pasteur pipettes (with bulbs)
- Masking tape or similar
- Permanent marker
- 2 red cabbage pH reference sheets (see image above)
- 1 plastic tote in which to put everything
Beforehand
1. Blend the red cabbage with some water in a blender, filter, transfer to a large bottle.
2. Make the coffee, tea, watered-down bleach etc, and transfer all those things to bottles or jars (these are the test samples)
3. Put everything in the tote
At event
1. Put ~1ml red cabbage juice in each test tube, put tubes in racks (these are the test vials)
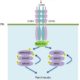
2. Put the various test samples in solo cups and equip each with a pipette (see photo below)
3. Put the weigh boats in front of each solo cup, each containing (if appropriate/possible) the raw ingredient in the solution contained in the corresponding solo cup (see photo below)
4. Use guided inquiry to help participants identify each test sample, then explain that the goal of the activity is to determine the pH of each test sample by using the test vials and the reference sheets. For younger participants it can work well to make it into a game by asking them to identify the most basic or most acidic test sample. Sometimes they get really excited and just want to mix lots of things together and make pretty colors 🙂
5. Give participants one test vial for each test sample and set them loose!

* Though the bleach should initially register as a very basic/alkaline solution, after a few seconds it may render the solution transparent as it oxidizes the color molecules in the tube.