Defense, fast and slow: activation of different MAPK pathways in response to wounding
In a world full of herbivores and mechanical stresses, the ability to sense and respond to wounding is crucial for plants, as injuries open doors for phytopathogen entry and allow uncontrolled evaporation. Wounding triggers local and systemic defenses that heal injuries, make plants taste unpleasant or toxic for herbivores or attract their natural enemies, and prime plant immunity to counteract pathogen invasion (Savatin et al., 2014). Wound responses are achieved by activation of signaling pathways that involve protein phosphorylation, jasmonic acid (JA) and ethylene production, as well as reprogramming of gene expression. Although mitogen-activated protein (MAP) kinase cascades, comprised of MAP kinase kinase kinases (MAP3Ks) that phosphorylate MAP kinase kinases (MKKs) that in turn activate MAP kinases (MPKs), are well-known signal transducers in defense against insects (Hettenhausen et al., 2015), the roles of specific MAPK modules in activating wound responses need further study. Here, Sözen et al. (2020) show that wounding induces a MAPK signaling module involving little known clade III MAP3Ks, the atypical MKK3 and the clade C MAPKs MPK1, MPK2 and MPK7. This induction is slower than the better studied MPK3/MPK6 pathway, and unlike MPK3/MPK6 activation, is dependent on JA signaling.
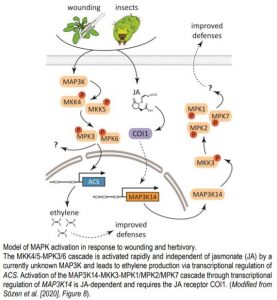 Clade C MAPKs MPK1 and MPK2 that act downstream of MKK3 had previously been associated with wounding and other stress responses (Ortiz-Masia et al., 2007). Sözen et al. tested clade C MPK activation in response to wounding and found that the activity of MPK1, MPK2, and MPK7 increased 30 min to 2 hours after wounding in an MKK3-dependent manner. The authors hypothesized that MKK3 is regulated by clade III MAP3Ks (MAP3K13-MAP3K20). In line with this, MKK3 interacted with MAP3K15-MAP3K20, as well as MAP3K13 and MAP3K14 (after truncation of the C-terminus) in a yeast two-hybrid assay; and both MKK3 and a clade III MAP3K were required for the activation of MPK2 transiently expressed in mkk3 protoplasts. MPK1/MPK2/MPK7 activation in response to wounding was dependent on de novo protein synthesis. Wounding led to a strong increase in MAP3K14 mRNA levels within 15-30 minutes, matching the timing of the relatively slow activation of MPK1/MPK2/MPK7 after wounding. The role of MAP3K14 as a regulator of MPK2 activation upon wounding was further supported by analysis of genome-edited plants encoding MAP3K14 versions with a truncated kinase domain. In the edited plants, MPK2 activation was strongly reduced.
Clade C MAPKs MPK1 and MPK2 that act downstream of MKK3 had previously been associated with wounding and other stress responses (Ortiz-Masia et al., 2007). Sözen et al. tested clade C MPK activation in response to wounding and found that the activity of MPK1, MPK2, and MPK7 increased 30 min to 2 hours after wounding in an MKK3-dependent manner. The authors hypothesized that MKK3 is regulated by clade III MAP3Ks (MAP3K13-MAP3K20). In line with this, MKK3 interacted with MAP3K15-MAP3K20, as well as MAP3K13 and MAP3K14 (after truncation of the C-terminus) in a yeast two-hybrid assay; and both MKK3 and a clade III MAP3K were required for the activation of MPK2 transiently expressed in mkk3 protoplasts. MPK1/MPK2/MPK7 activation in response to wounding was dependent on de novo protein synthesis. Wounding led to a strong increase in MAP3K14 mRNA levels within 15-30 minutes, matching the timing of the relatively slow activation of MPK1/MPK2/MPK7 after wounding. The role of MAP3K14 as a regulator of MPK2 activation upon wounding was further supported by analysis of genome-edited plants encoding MAP3K14 versions with a truncated kinase domain. In the edited plants, MPK2 activation was strongly reduced.
JA has a central role in wounding responses, so the authors tested the effect of JA on MPK2 activation and found that JA activated MPK2 in wild-type, but not in mkk3 plants with similar kinetics as wounding. In turn, in JA biosynthesis and signaling mutants, wounding-induced activation of MPK2 was strongly reduced, further supporting the importance of JA biosynthesis and signaling in the regulation of MKK3-MPK2 module in response to wounding.
In line with previous findings (Hettenhausen et al., 2015), the authors detected rapid MPK3 and MPK6 activation 15 minutes after wounding, but found that this occurred independent of MKK3 and MAP3K14 and dependent on MKK4 and MKK5. However, MPK3/MPK6 activation was not triggered by JA treatment and not compromised in a mutant deficient in JA perception, indicating JA independence of the MKK4/MKK5-MPK3/MPK6 module in wound responses.
Wounding assays only partly mimic herbivory, so the authors asked, whether insect feeding on plants triggers similar activation of the MAPK cascades. Feeding of Spodoptera littoralis larvae on plants induced weak activation of MPK3/MPK6 after 15 minutes, but strong activation of MPK2 after 1 hour, which was dependent on MKK3 and JA sensing. After long term feeding on wild-type or mkk3 plants, S. littoralis larvae grown on mkk3 plants were bigger, suggesting that the defense system in mkk3 did not efficiently deter them. Taken together, these data suggest that the relatively slow JA-dependent activation of the MAP3K14-MKK3-MPK1/MPK2/MPK7 signaling module has a prominent role in activating defenses to protect plants against herbivory (see figure). Identifying the downstream targets of the module will help to better understand how plants defend themselves against herbivores.
Hanna Hõrak
Institute of Technology
University of Tartu, Estonia
ORCID: 0000-0002-6392-859X
REFERENCES
Hettenhausen, C., Schuman, M.C., and Wu, J. (2015). MAPK signaling – a key element in plant defense response to insects. Insect Sci 22: 157–164.
Ortiz-Masia, D., Perez-Amador, M.A., Carbonell, J., and Marcote, M.J. (2007). Diverse stress signals activate the C1 subgroup MAP kinases of Arabidopsis. FEBS Letters 581: 1834–1840.
Savatin, D.V., Gramegna, G., Modesti, V., and Cervone, F. (2014). Wounding in the plant tissue: the defense of a dangerous passage. Front. Plant Sci. 5:470.
Sözen, C., Schenk, S.T., Boudsocq, M., Chardin, C., Almeida-Trapp, M., Krapp, A., Hirt, H., Mithöfer, A., and Colcombet, J. (2020). Wounding and Insect Feeding Trigger two Independent MAPK Pathways with Distinct Regulation and Kinetics. The Plant Cell. Published April 2020. DOI: https://doi.org/10.1105/tpc.19.00917