Callose Suppresses Low Calcium-Induced Cell Death
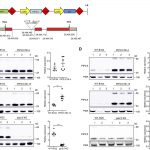
Calcium (Ca) deficiency symptoms often occur in crops because Ca is mainly translocated by the transpiration stream, and conditions that affect transpiration can cause Ca deficiency. Because Ca is translocated via the transpiration stream, it tends to accumulate more in older, 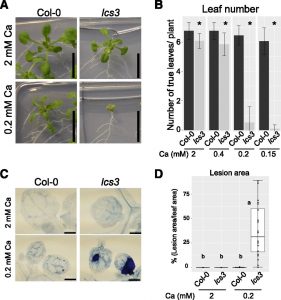 expanded leaves and less in rapidly growing tissues such as new, small leaves, where the demand for Ca to provide new cell wall materials is high. The Ca deficiency symptoms occur in rapidly growing tissues such as new leaves (tip-burn) and fruits (blossom end-rot), which is thought to be caused by an imbalance between the supply of and demand for Ca among tissues. Shikanai et al. (10.1104/pp.19.00784) now shed light on the critical involvement of callose synthesis in the survival of Arabidopsis (Arabidopsis thaliana) under low-Ca conditions. A mutant sensitive to low-Ca conditions, low calcium sensitive3 (lcs3), exhibited high levels of cell death in emerging leaves and had defects in its expanding true leaves under low-Ca conditions. Further analyses showed that the causal mutation was located in a putative b-1,3-glucan (callose) synthase gene, GLUCAN SYNTHASE-LIKE10 (GSL10). Yeast complementation assay results showed that GSL10 encodes a functional callose synthase. Ectopic callose significantly accumulated in wild-type plants under low-Ca conditions, but at a low level in lcs3. The phenotype of lcs3, which included failure of leaf expansion, cell death, and reduced ectopic callose levels under low-Ca conditions, was similar to the effects of callose synthase inhibitors on wild-type plants. These results suggest that callose synthesis plays a key role in plants’ survival strategies under low-Ca conditions.
expanded leaves and less in rapidly growing tissues such as new, small leaves, where the demand for Ca to provide new cell wall materials is high. The Ca deficiency symptoms occur in rapidly growing tissues such as new leaves (tip-burn) and fruits (blossom end-rot), which is thought to be caused by an imbalance between the supply of and demand for Ca among tissues. Shikanai et al. (10.1104/pp.19.00784) now shed light on the critical involvement of callose synthesis in the survival of Arabidopsis (Arabidopsis thaliana) under low-Ca conditions. A mutant sensitive to low-Ca conditions, low calcium sensitive3 (lcs3), exhibited high levels of cell death in emerging leaves and had defects in its expanding true leaves under low-Ca conditions. Further analyses showed that the causal mutation was located in a putative b-1,3-glucan (callose) synthase gene, GLUCAN SYNTHASE-LIKE10 (GSL10). Yeast complementation assay results showed that GSL10 encodes a functional callose synthase. Ectopic callose significantly accumulated in wild-type plants under low-Ca conditions, but at a low level in lcs3. The phenotype of lcs3, which included failure of leaf expansion, cell death, and reduced ectopic callose levels under low-Ca conditions, was similar to the effects of callose synthase inhibitors on wild-type plants. These results suggest that callose synthesis plays a key role in plants’ survival strategies under low-Ca conditions.