The Fate of Acetate During Hypoxia
Under hypoxia, Acetyl-CoA Synthetase (ACS), an enzyme that synthesizes acetyl CoA, recovers carbon that would otherwise be lost from the 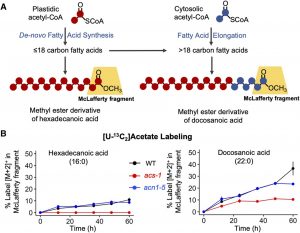 plant as ethanol. Plastid-localized ACS metabolizes cellular acetate and contributes to the de novo biosynthesis of fatty acids and Leu. On the other hand, a peroxisome-localized enzyme ACETATE NON-UTILIZING1 (ACN1) ACN1 enables the incorporation of acetate into organic acids and amino acids. Thus, the activation of acetate in distinct subcellular compartments provides plants with the metabolic flexibility necessary to maintain physiological levels of acetate. Because the acetyl-CoA metabolic network is highly compartmentalized in eukaryotic cells, there is a need to better understand how acetyl-CoA generation and utilization are genetically and biochemically coordinated among different cellular and subcellular compartment. Fu et al. (10.1104/pp.19.01162) have examined the functions and interplay of ACS and ACN1 in acetate/acetyl-CoA metabolism in Arabidopsis. Using multiple mutant stocks and stable isotope-assisted metabolic analyses, the authors demonstrate that plastidic ACS and peroxisomal ACN1 provide redundant functions to prevent hyperaccumulation of acetate. In contrast to the near wild-type morphological and metabolic phenotypes of acs or acn1 mutants, the acs acn1 double mutant is delayed in growth and sterile, which is associated with hyperaccumulation of cellular acetate and decreased accumulation of acetyl-CoA-derived intermediates of central metabolism.
plant as ethanol. Plastid-localized ACS metabolizes cellular acetate and contributes to the de novo biosynthesis of fatty acids and Leu. On the other hand, a peroxisome-localized enzyme ACETATE NON-UTILIZING1 (ACN1) ACN1 enables the incorporation of acetate into organic acids and amino acids. Thus, the activation of acetate in distinct subcellular compartments provides plants with the metabolic flexibility necessary to maintain physiological levels of acetate. Because the acetyl-CoA metabolic network is highly compartmentalized in eukaryotic cells, there is a need to better understand how acetyl-CoA generation and utilization are genetically and biochemically coordinated among different cellular and subcellular compartment. Fu et al. (10.1104/pp.19.01162) have examined the functions and interplay of ACS and ACN1 in acetate/acetyl-CoA metabolism in Arabidopsis. Using multiple mutant stocks and stable isotope-assisted metabolic analyses, the authors demonstrate that plastidic ACS and peroxisomal ACN1 provide redundant functions to prevent hyperaccumulation of acetate. In contrast to the near wild-type morphological and metabolic phenotypes of acs or acn1 mutants, the acs acn1 double mutant is delayed in growth and sterile, which is associated with hyperaccumulation of cellular acetate and decreased accumulation of acetyl-CoA-derived intermediates of central metabolism.



