Systems Analysis of Lignin Mutants
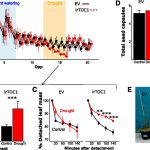
Lignin is a complex polymer deposited in plant cell walls that provides mechanical support and facilitates the transport of water and solutes through 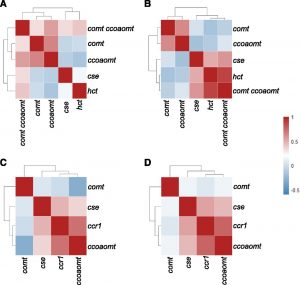 the vascular system, and aids in plant defense. Lignin waterproofs plant cells by providing a hydrophobic environment by chemical bonding with cellulose and hemicellulose. This lignin-polysaccharide matrix underlies the recalcitrance that limits chemical, enzymatic, and microbial digestion of plant cell walls, thereby hindering the conversion of lignocellulosic biomass to liquid biofuels. Genetic modification of the lignin pathway, however, can result in diverse and unpredictable impacts on plant physiology, especially the development of dwarf plants. In spite of extensive studies on individual dwarf lignin mutants in several different species, there is no clear agreement as to whether a common mechanism is responsible for these dwarf phenotypes. To address this question, Ha et al. (10.1104/pp.19.00533) have analyzed transposon insertion various single mutants of Medicago truncatula that have specific defects in the monolignol pathway, as well as one double mutant. These plants exhibit growth phenotypes from essentially wild type to severely retarded. Extensive phenotypic, transcriptomic, and metabolomics analyses, including structural characterization of differentially expressed compounds, revealed diverse phenotypic consequences of lignin pathway. Notable phenotypes among the mutants with severe growth impairment were increased trichome numbers, accumulation of a variety of triterpene saponins, and extensive but differential ectopic expression of defense response genes. The authors propose that reallocation of resources into defense pathways is linked to the severity of the final growth phenotype in monolignol pathway mutants of M. truncatula, although it remains unclear whether this is a cause or an effect of the growth impairment.
the vascular system, and aids in plant defense. Lignin waterproofs plant cells by providing a hydrophobic environment by chemical bonding with cellulose and hemicellulose. This lignin-polysaccharide matrix underlies the recalcitrance that limits chemical, enzymatic, and microbial digestion of plant cell walls, thereby hindering the conversion of lignocellulosic biomass to liquid biofuels. Genetic modification of the lignin pathway, however, can result in diverse and unpredictable impacts on plant physiology, especially the development of dwarf plants. In spite of extensive studies on individual dwarf lignin mutants in several different species, there is no clear agreement as to whether a common mechanism is responsible for these dwarf phenotypes. To address this question, Ha et al. (10.1104/pp.19.00533) have analyzed transposon insertion various single mutants of Medicago truncatula that have specific defects in the monolignol pathway, as well as one double mutant. These plants exhibit growth phenotypes from essentially wild type to severely retarded. Extensive phenotypic, transcriptomic, and metabolomics analyses, including structural characterization of differentially expressed compounds, revealed diverse phenotypic consequences of lignin pathway. Notable phenotypes among the mutants with severe growth impairment were increased trichome numbers, accumulation of a variety of triterpene saponins, and extensive but differential ectopic expression of defense response genes. The authors propose that reallocation of resources into defense pathways is linked to the severity of the final growth phenotype in monolignol pathway mutants of M. truncatula, although it remains unclear whether this is a cause or an effect of the growth impairment.