Setting Time for a Hot Date: Paused Embryo Development and Protective Organogenesis Allow Dates to Cope with the Desert Environment
Adaptive developmental plasticity, in which changing environmental conditions modulate morphogenesis, can help organisms survive harsh conditions. Common examples include the protection of shoot apical meristems by transient arrest and sequestration into bud-like structures in wintering evergreens (e.g. Ruttink et al., 2007), embryonic diapause in mammals, and dormancy of seeds containing fully developed embryos.
Date palm (Phoenix dactylifera L.) is one of the few fruit tree species capable of surviving in desert environments, and as such, date palms are essential for desert agricultural production. In a new study, Xiao and colleagues (2019) investigated several mechanisms that allow date palms to survive the extreme and highly variable temperatures and drought conditions of the desert.
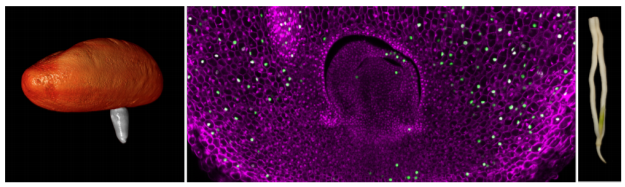
Germination of many seed plants involves the emergence of distinct root and then shoot structures (cotyledons or leaves). By contrast, date palms undergo a germination mode referred to as remote tubular germination that involves the initial emergence of a root-like structure termed the cotyledonary petiole (CP). Subsequently, the CP elongates and thickens, and at later stages of germination the primary leaf emerges from an opening in the CP. The distal CP tip further elongates to form the main root.
Xiao et al. found that the embryo within the CP tip during the first few weeks post-germination showed high anatomical similarity to the embryo found in pre-germinated seeds, suggesting a state of paused embryonic development during early germination (see Figure). This pause was supported by low rates of cell division, monitored by ethynyl deoxyuridine (EdU) incorporation into DNA, low expression of developmental genes and increase of hormones involved in dormancy.
 In later stages of the germination process, embryo development continues. Within the CP, the apical pole of the developing seedling shows shoot apical meristem like features, while the external distal tip of the CP develops root-like features such as accumulation of starch granules, and genes marking the differentiated columella layers. The paused embryonic development, followed by protected organogenesis within the CP, suggests that date palms have evolved a developmental program that aims to provide maximum protection to the future seedlings from the desert environment.
In later stages of the germination process, embryo development continues. Within the CP, the apical pole of the developing seedling shows shoot apical meristem like features, while the external distal tip of the CP develops root-like features such as accumulation of starch granules, and genes marking the differentiated columella layers. The paused embryonic development, followed by protected organogenesis within the CP, suggests that date palms have evolved a developmental program that aims to provide maximum protection to the future seedlings from the desert environment.
Post-germination, date palms develop an intricate root system consisting of vertically growing main and anchor roots, secondary lateral roots, and agravitropic pneumatophores. Main roots showed a complex differential accumulation of suberin and lignin which may prevent water loss and balance ion fluxes. Root transcriptomic data revealed high expression aquaporins, facilitators of water uptake, as well as genes involved in bacterial responses in roots, the latter of which reflects a role for root bacterial communities in adaptation to stress conditions. Finally, pneumatophores were shown to have differential root thickness along the proximal-distal axis, arising from both difference in cell sizes and numbers. Although the function of pneumatophores remains unclear, the authors propose a role in increasing water uptake.
Together, these data support multiple adaptive strategies used by date palms to survive harsh desert environments, including pausing of embryonic development, protection of meristematic tissues during early organogenesis, and establishment of specialized root systems.
Ruttink T, Arend M, Morreel K, Storme V, Rombauts S, Fromm J., Bhalerao RP, Boerjan W, and Rohde A (2007). A molecular timetable for apical bud formation and dormancy induction in poplar. Plant Cell 19: 2370-2390.
Xiao TT, Aragón Raygoza A, Caballero Pérez J, Kirschner G, Deng Y, Atkinson B, Sturrock C, Lube, V, Wang JY, Lubineau G, al Babili S, Cruz Ramírez A, Bennett M and Blilou I. (2019). Emergent protective organogenesis in date palms: A morpho-devo-dynamic adaptive strategy during early development. Plant Cell https://doi.org/10.1105/tpc.19.00008.
Figure legend:
Germination in date palms. Germination begins with the emergence of a root-like structure termed the cotyledonary petiole (CP; left panel). Xiao et al. reveal that in date palm, the embryo pauses development at this stage (middle panel; monitored by confocal microscopy and EdU staining of dividing cells [green]). At later stages, this developmental pause is lifted and organogenesis continue within the CP, ultimately resulting in emergence of leaves (right panel). This mode of remote germination likely helps date palms survive in harsh desert environments. (Figure courtesy of T.T. Xiao, V. Lube, and I. Blilou.)