Say “Ah!” The Right Amounts of Brassinosteroids and Hydrogen Peroxide Open the “Mouths” of Plant Leaves
Stomata, from the Greek “mouths,” are tiny pores in the epidermis of many plant aerial tissues that are “fed” carbon dioxide for making sugar. But plants also lose water through these same pores via transpiration. Stomatal aperture regulation in response to environmental cues is therefore of fundamental importance.
Changes in the turgor pressure of guard cells that flank stomatal pores bring about stomatal pore size alterations. One key to stomatal opening is known to be the degradation of starch in the guard cells, in part accomplished by β-AMYLASE1 (BAM1) and α-AMYLASE3 (AMY3) enzymes. Double–knock-out bam1 amy3 mutants accumulate starch and fail to fully open their stomata in response to light exposure at the beginning of the day. But what regulates this process?
 Li et al. (2020) tackle the complex topic of hormone and reactive oxygen species (ROS) influences on stomatal aperture. It is well known that the hormone abscisic acid functions in stomatal closure, in particular in response to drought. However, the role of brassinosteroid (BR) hormones in regulating stomatal aperture is less straightforward. Unsurprisingly, as with most hormones, the effects are dosage dependent. But at least in the case of stomatal aperture regulation in Arabidopsis (Arabidopsis thaliana), there is not simply a dose at which the effect of BR plateaus with increasing concentrations; rather, opposing effects occur depending on dosage: a little bit of BR opens stomata, but a lot of BR closes stomata. Hydrogen peroxide (H2O2), once thought of only as a ROS that damages cells, is now known to be a second messenger of BR signaling, and follows a similar dosage-effect pattern: low concentrations of H2O2 open stomata, while high concentrations close them.
Li et al. (2020) tackle the complex topic of hormone and reactive oxygen species (ROS) influences on stomatal aperture. It is well known that the hormone abscisic acid functions in stomatal closure, in particular in response to drought. However, the role of brassinosteroid (BR) hormones in regulating stomatal aperture is less straightforward. Unsurprisingly, as with most hormones, the effects are dosage dependent. But at least in the case of stomatal aperture regulation in Arabidopsis (Arabidopsis thaliana), there is not simply a dose at which the effect of BR plateaus with increasing concentrations; rather, opposing effects occur depending on dosage: a little bit of BR opens stomata, but a lot of BR closes stomata. Hydrogen peroxide (H2O2), once thought of only as a ROS that damages cells, is now known to be a second messenger of BR signaling, and follows a similar dosage-effect pattern: low concentrations of H2O2 open stomata, while high concentrations close them.
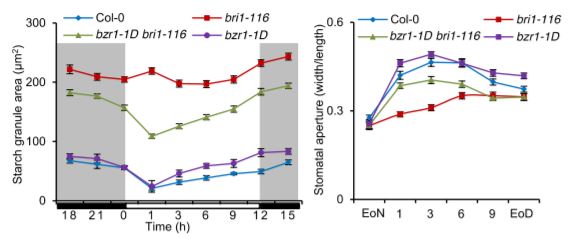
A clear and consistent pattern emerged in the Li et al. (2020) study, whereby guard-cell starch levels inversely correlated with stomatal aperture in several different mutant and transgenic plants, including those affecting BR levels. For example, the BR-insensitive bri1-116 and BR-deficient det2 mutants displayed both increased guard-cell starch levels and decreased stomatal apertures in response to light as compared to those of wild-type plants. By contrast, the dominant gain-of-function bzr1-1D mutant, in which BR signaling is continually activated, displayed decreased guard-cell starch levels and increased stomatal apertures similar to those of wild-type plants, and the mutation was able to partially suppress the guard-cell starch and stomatal aperture phenotypes of the bri1-116 and det2 mutants (see figure).
Additionally, unlike in wild-type plants, treatments with either BR or H2O2 were unable to cause an increase in starch degradation and a corresponding increase in stomatal aperture in plants overexpressing the CATALASE2 gene, whose enzyme product breaks down H2O2. Furthermore, the interaction between the transcription factors GBF2 and BZR1, which increase BAM1 expression, was enhanced by H2O2. Stomatal aperture was reduced and was less responsive to BR or H2O2 in gbf2-1 mutant plants, and was increased in plants overexpressing GBF2. Finally, BAM1 expression in guard cells was increased in response to BR or H2O2, and its transcripts were reduced in bri1-116, det2, and gbf2-1 mutants but increased in the bzr1-1D mutant. These results point to a model in which BR signaling in guard cells increases H2O2 to enhance GBF2 and BZR1 interaction, which in turn increases BAM1 expression, resulting in starch degradation, followed by stomatal opening.
Intriguingly and perhaps counterintuitively, Yamauchi et al. (2019) recently reported that increased ROS in guard cells causes stomatal closing, which is alleviated by autophagy of oxidized peroxisomes (“pexophagy”). However, because the levels of H2O2 in guard cells cannot be precisely measured, the decrease in stomatal aperture in response to increased ROS/H2O2 levels may not be irreconcilable with the current Li et al. (2020) study. Once the complex interplay of hormones, starch degradation, and pexophagy involved in stomatal aperture regulation is solved, stomata may come to mind when our mouths utter “Ah!”
Anne C. Rea
Michigan State University
MSU-DOE Plant Research Laboratory
ORCID: 0000-0002-2996-5709
REFERENCES
Li, J.-G., Fan, M., Hua, W., Tian, Y., Chen, L.-G., Sun, Y., and Bai, M.-Y. (2020). Brassinosteroid and hydrogen peroxide interdependently induce stomatal opening by promoting guard cell starch degradation. Plant Cell DOI: https://doi.org/10.1105/tpc.19.00587.
Yamauchi, S., Mano, S., Oikawa, K., Hikino, K., Teshima, K.M., Kimori, Y., Nishimura, M., Shimazaki, K., and Takemiya, A. (2019). Autophagy controls reactive oxygen species homeostasis in guard cells that is essential for stomatal opening. Proc. Natl. Acad. Sci. U.S.A. 116: 19187–19192.