Salt stress disturbs sugar metabolism
Zheng-Wei Fu (State Key Laboratory of Hybrid Rice, College of Life Sciences, Renmin Hospital of Wuhan University, Wuhan University, Wuhan, China)
Background: Soil salinization is a serious environmental threat to global agriculture. High salinity results in osmotic stress, ionic stress, and secondary stresses including oxidative stress caused by ROS (reactive oxygen species). Most studies on plant salt stress response have focused on how plants maintain cellular osmotic, ionic, and ROS homeostasis to adapt to salt stress. But how plants are damaged by salt stress is poorly understood. While hydrogen peroxide (H2O2), a major form of ROS, can oxidize proteins to alter their functions, how salt stress-indued H2O2 inhibits plant growth and development remains elusive.
Question: What pathway functions downstream of H2O2 and which functional proteins are modified by H2O2 in salt-stressed plants?
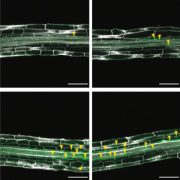
Findings: We found that plastid triose phosphate isomerase (pdTPI), a key enzyme of sugar metabolism, confers plant salt stress tolerance. Salt stress–induced H2O2 inhibits pdTPI activity to disturb metabolic processes, thus promoting the accumulation of a sugar metabolite, methylglyoxal (MG). We also showed that MG is a key factor limiting plant growth and in turn induces H2O2 production. Interestingly, targeting a H2O2 degrading enzyme originally located in peroxisomes to chloroplasts can prevent salt-induced over-accumulation of H2O2 and confer salt stress tolerance. In addition, pdTPI can be modified by H2O2 in salt-stressed plants, implying that H2O2 inhibits pdTPI activity by directly oxidizing it.
 Next steps: In future studies, we will investigate whether salt stress–induced H2O2 inhibits pdTPI activity by directly oxidizing it. We will also focus on whether and how MG inhibits plant growth by modifying proteins.
Next steps: In future studies, we will investigate whether salt stress–induced H2O2 inhibits pdTPI activity by directly oxidizing it. We will also focus on whether and how MG inhibits plant growth by modifying proteins.
Reference:
Zheng-Wei Fu, Yu-Rui Feng, Xiang Gao, Feng Ding, Jian-Hui Li, Ting-Ting Yuan, and Ying-Tang Lu (2023) Salt stress-induced chloroplastic hydrogen peroxide stimulates pdTPI sulfenylation and methylglyoxal accumulation. https://doi.org/10.1093/plcell/koad019