Root Foraging under Low Nitrogen Depends on Brassinosteroids
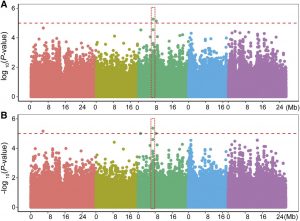 Plant root systems alter their architecture upon encountering spatial heterogeneity in the nutrient concentrations of the soil. For example, when roots are subjected to decreasing N availability that causes mild N deficiency, lateral root emergence increases moderately, whereas elongation of primary and lateral roots is strongly induced. These architectural modifications result in a systemic foraging response that increases soil exploration by an expanded root system but at the cost of a lower root diameter and lower cell wall stability. The mechanisms underlying this foraging response, however, remain poorly understood. Jia et al. (10.1104/pp.20.00440) have performed a genome-wide association study in Arabidopsis and show that noncoding variations of the brassinosteroid (BR) biosynthesis gene DWARF1 (DWF1) lead to variations of the DWF1 transcript level that contributes to natural variation of root elongation under low N. In addition to DWF1, other central BR biosynthesis genes are also upregulated under low N. Knockout and knockdown mutants of these genes showed significant reductions in their root elongation responses to low N. The authors also show that low-N-induced root elongation is associated with above-ground N content and that the overexpression of DWF1 significantly improves plant growth and overall N accumulation. Thus, mild N deficiency induces key genes in BR biosynthesis and natural variation in BR synthesis contributes to the root foraging response..
Plant root systems alter their architecture upon encountering spatial heterogeneity in the nutrient concentrations of the soil. For example, when roots are subjected to decreasing N availability that causes mild N deficiency, lateral root emergence increases moderately, whereas elongation of primary and lateral roots is strongly induced. These architectural modifications result in a systemic foraging response that increases soil exploration by an expanded root system but at the cost of a lower root diameter and lower cell wall stability. The mechanisms underlying this foraging response, however, remain poorly understood. Jia et al. (10.1104/pp.20.00440) have performed a genome-wide association study in Arabidopsis and show that noncoding variations of the brassinosteroid (BR) biosynthesis gene DWARF1 (DWF1) lead to variations of the DWF1 transcript level that contributes to natural variation of root elongation under low N. In addition to DWF1, other central BR biosynthesis genes are also upregulated under low N. Knockout and knockdown mutants of these genes showed significant reductions in their root elongation responses to low N. The authors also show that low-N-induced root elongation is associated with above-ground N content and that the overexpression of DWF1 significantly improves plant growth and overall N accumulation. Thus, mild N deficiency induces key genes in BR biosynthesis and natural variation in BR synthesis contributes to the root foraging response..



