Fine-tuning plant growth in the face of drought
IN BRIEF by Kathleen L. Farquharson kfarquharson@aspb.org
 Limiting shoot growth is an important survival strategy for plants during times of drought; smaller leaves mean that less water is lost through transpiration and more is retained in the soil. As drought stress restricts both cell division and expansion – processes that rely on specialized microtubule arrays – factors that regulate microtubule organization and stability likely affect a plant’s ability to grow in dry conditions. However, the signaling pathways that link drought stress with changes in microtubule organization are unknown.
Limiting shoot growth is an important survival strategy for plants during times of drought; smaller leaves mean that less water is lost through transpiration and more is retained in the soil. As drought stress restricts both cell division and expansion – processes that rely on specialized microtubule arrays – factors that regulate microtubule organization and stability likely affect a plant’s ability to grow in dry conditions. However, the signaling pathways that link drought stress with changes in microtubule organization are unknown.
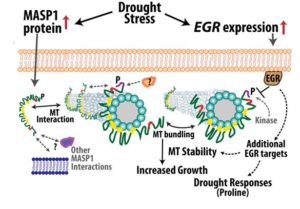
Now, Bhaskara et al. (2017) report a signaling module in Arabidopsis thaliana that influences microtubule organization and regulates plant growth, particularly during prolonged exposure to drought stress. Prompted by the finding that several Type 2C protein phosphatases (PP2Cs) function in drought signaling (Bhaskara et al., 2012; Cutler et al., 2010; Schweighofer, 2004), the authors identified three closely related PPC2s (EGRs) that inhibited plant growth and were dramatically induced by drought stress. Phosphoproteomic analysis revealed 17 putative EGR targets, many of which had established roles in cytoskeleton regulation or vesicle trafficking. The authors focused on one of these targets [which they named Microtubule Associated Stress Protein 1 (MASP1)], due to its massive increase in abundance in drought-stressed plants.
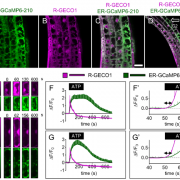
In contrast to the EGR protein phosphatases, MASP1 was found to promote growth, both under control and drought stress conditions. The authors showed that MASP1 associated with EGR proteins at the cell periphery. Given the enrichment of cytoskeleton-related proteins among the putative EGR targets, the authors then used lines expressing GFP-tagged a-tubulin to examine if EGR and MASP1 regulate microtubule organization. In agreement with previous reports (Mei et al., 2012 and Fujita et al., 2013), the authors found that cortical microtubule arrays became disorganized within a few hours of exposure to drought stress and partially recovered over the next few days. Strikingly, plants that overexpressed MASP1 or lacked EGR phosphatases had more stable microtubule arrays and exhibited more rapid recovery of microtubule organization under drought stress conditions. Further analysis showed that MASP1 associates with microtubules in vitro. In planta, MASP1 phosphorylation at S670 is critical for growth promotion and microtubule stabilization. Finally, epistasis tests indicated that EGRs and MASP1 function in a common pathway, with MASP1 acting downstream of EGRs.
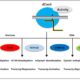
Based on these findings, the authors developed a model in which EGR phosphatases and MASP1 regulate microtubule stability during drought stress, with the former reducing and the latter promoting microtubule stability and anisotropic cell expansion (see figure). Fine-tuning the activity of these two opposing sets of proteins may improve plant growth and yield during moderate drought.
References
Bhaskara, G.B., Nguyen, T.T., Verslues, P.E. (2012) Unique drought resistance functions of the highly ABA-induced clade A protein phosphatase 2Cs. Plant Physiol. 160: 379-395.
Bhaskara, G.B., Wen, T.-N., Nguyen, T.T., Verslues, P.E. Protein Phosphatase 2Cs and Microtubule-Associated Stress Protein 1 Control Microtubule Stability, Plant Growth, and Drought Response. Plant Cell 10.1105/tpc.116.00847.
Cutler, S.R., Rodriguez, P.L., Finkelstein, R.R., and Abrams, S.R. (2010). Abscisic Acid: Emergence of a Core Signaling Network. Annual Review of Plant Biology, 61: 651-679.
Fujita, S., Pytela, J., Hotta, T., Kato, T., Hamada, T., Akamatsu, R., Ishida, Y., Kutsuna, N., Hasezawa, S., Nomura, Y., Nakagami, H., and Hashimoto, T. (2013). An Atypical Tubulin Kinase Mediates Stress-Induced Microtubule Depolymerization in Arabidopsis. Current Biology 23: 1969-1978.
Mei, Y., Gao, H.B., Yuan, M., and Xue, H.W. (2012). The Arabidopsis ARCP Protein, CSI1, Which Is Required for Microtubule Stability, Is Necessary for Root and Anther Development. Plant Cell 24: 1066-1080.
Schweighofer, A., Hirt, H., and Meskiene, L. (2004). Plant PP2C Phosphatases: Emerging Functions in Stress Signaling. Trends in Plant Science 9: 236-243.





Leave a Reply
Want to join the discussion?Feel free to contribute!