Overcoming Algal Vitamin B12 Auxotrophy by Experimental Evolution
Vitamin B12 (B12) is produced only by prokaryotes (Fang et al., 2017), but is also widely required among eukaryotes as an enzymatic cofactor. In the green lineage, the only B12-dependent enzyme is methionine synthase. Several green algae can synthesize methionine only thanks to B12 provided by mutualistic prokaryotes (Croft et al., 2005, Grant et al., 2014) and require B12 supplementation for axenic growth in vitro. However, some green algae and all land plants possess an alternative, B12-independent methionine synthase and therefore do not require B12 for growth. Furthermore, other green algae, including the model organism Chlamydomonas reinhardtii, carry both methionine synthase isoforms and regulate their expression according to B12 availability (Helliwell et al., 2014). Algal B12 auxotrophy is heterogeneously distributed and is believed to have independently evolved many times (Helliwell et al., 2011).
 Recently, Helliwell and colleagues (2015) artificially evolved a stable B12 auxotrophic C. reinhardtii strain, the metE7 strain, which relies only on the B12-dependent methionine synthase for growth because it contains a portion of a transposon inside the B12-independent methionine synthase gene. Now, in this issue of Plant Physiology, Bunbury at al. (2020) take advantage of the B12 auxotrophic C. reinhardtii metE7 strain to investigate the role of B12 in algal metabolism and growth, as well as evolutionary strategies to withstand B12 scarcity.
Recently, Helliwell and colleagues (2015) artificially evolved a stable B12 auxotrophic C. reinhardtii strain, the metE7 strain, which relies only on the B12-dependent methionine synthase for growth because it contains a portion of a transposon inside the B12-independent methionine synthase gene. Now, in this issue of Plant Physiology, Bunbury at al. (2020) take advantage of the B12 auxotrophic C. reinhardtii metE7 strain to investigate the role of B12 in algal metabolism and growth, as well as evolutionary strategies to withstand B12 scarcity.
First, the authors noted that the metE7 strain displays various, serious symptoms of stress when cultured in the absence of B12, including impaired cell division, reduced viability, low photosynthetic efficiency and impaired metabolism, with a decline in the levels of chlorophyll, proteins, polar lipids and free fatty acids, but accumulation of starch and triacylglycerides. Those changes mimic some consequences of nitrogen deprivation in wild-type C. reinhardtii. Surprisingly, nitrogen deprivation itself was found to improve metE7 viability. One important aspect of combining nitrogen and B12 deprivation appeared to be a reduction in the accumulation of reactive oxygen species (ROS), suggesting that a ROS decrease may be one factor linking nitrogen deprivation to reduced cell death when B12 is scarce.
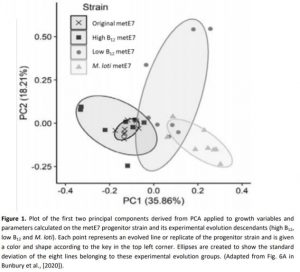
To determine if metE7 could evolve improved tolerance to B12 limiting conditions, Bunbury at al. (2020) set up a ten-month artificial evolution experiment in which metE7 was grown in the presence of: i) high B12 levels (1000 ng/l), ii) low B12 levels (25 ng/l), or iii) no B12 supplementation but in co-culture with the bacterium Mesorhizobium loti (Fig. 1). At the end of the experiment, algae co-cultured with bacteria were capable of higher B12 use efficiency and reached higher optical density at small doses of B12 in comparison to the original strain. In addition, viability at low B12 concentration and survival in B12 deprivation were improved for both the co-cultured and low-B12-cultured metE7 strain. These results demonstrate that even a newly evolved B12 auxotroph can quickly develop strategies to improve its fitness in suboptimal conditions, including the establishment of trophic bacterial partnerships. Growth data suggested that the reduction of the growth rate is a necessary trade-off in adaptation to B12 auxotrophy.
Overall, this article advances our understanding of metabolic interactions between various pathways and underlines the importance of ecological relationships for nutrient supply in shaping evolution. A follow-up investigation on metabolic, immunity and epigenetic changes that occurred in the metE7-M. loti ten-month co-culture will potentially pinpoint early mechanisms in the establishment of algal–bacterial symbioses.
Elisa Dell’Aglio, INSA-Lyon, 69100 Villeurbanne, France. ORCID ID: 0000-0001-6363-9109
REFERENCES
Bunbury F., Helliwell K.E., Mehrshahi P., Davey MP, Salmon D, Holzer A, Smirnoff N, Smith AG (2020). Responses of a Newly Evolved Auxotroph of Chlamydomonas to B 12 Deprivation. Plant Physiology https://doi.org/10.1104/pp.19.01375
Croft MT, Lawrence AD, Raux-Deery E, Warren MJ, Smith AG (2005) Algae acquire vitamin B 12 through a symbiotic relationship with bacteria. Nature 438:90-93.
Fang H, Jie K, Dawei Z (2017) Microbial production of vitamin B 12: a review and future perspectives. Microbial cell factories 16(1):15.
Grant MA, Kazamia E, Cicuta P, Smith AG (2014) Direct exchange of vitamin B 12 is demonstrated by modelling the growth dynamics of algal–bacterial cocultures. The ISME journal 8:1418-1427.
Helliwell KE, Wheeler GL, Leptos KC, Goldstein RE, Smith AG (2011) Insights into the evolution of vitamin B12 auxotrophy from sequenced algal genomes. Molecular biology and evolution 28:2921-2933.
Helliwell KE, Scaife MA, Sasso S, Araujo APU, Purton S, Smith AG (2014) Unraveling vitamin B12-responsive gene regulation in algae. Plant physiology 165:388-397.
Helliwell KE, Collins S, Kazamia E, Purton S, Wheeler GL, Smith AG (2015) Fundamental shift in vitamin B 12 eco-physiology of a model alga demonstrated by experimental evolution. The ISME journal 9:1446-1455.



