On the Inside: Cell Wall Acetylation
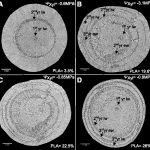
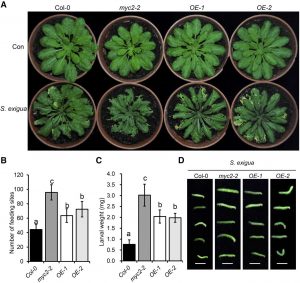 In plant cells, cellulose microfibrils make up the basic skeleton of the rigid cell wall while hemicellulose and pectin fill the space between cellulose microfibrils. Cell walls constitute the first wall in plant defense against pathogens and cell wall polysaccharides, and their acetylation status greatly affects responses to pathogens. Many cell wall polysaccharides undergo extensive O-acetylation, imparting important physiochemical properties to the polymers. The O-acetylation of polysaccharides modifies plant cell walls by changing their physicochemical properties and, consequently, their structure and function. In this report, Sun et al. (10.1104/pp.20.00683) cloned a previously identified stunted growth mutant named sucrose uncoupled1 (sun1) in Arabidopsis (Arabidopsis thaliana). SUN1 encodes a member of the TRICHOME BIREFRINGEN-LIKE family, AtTBL37. TBL37 (SUN1) is highly expressed in fast-growing plant tissues and encodes a Golgi apparatus-localized protein that regulates secondary cell wall thickening and acetylation. In tbl37-1, jasmonate signaling and the expression of downstream chemical defense genes, are increased but, cell wall acetylation is reduced, resulting in stunted growth, collapsed xylem vessels, and increased susceptibility to herbivorous insects. A key transcription factor involved in jasmonate signaling, MYC2, binds to and induces AtTBL37 expression and also enhances cell wall acetylation. The mutant myc2-2 is as susceptible to insect feeding as tbl37-1. The overexpression of AtTBL37 in a myc2-2 background increases resistance to herbivory. These findings reveal that AtTBL37-mediated acetylation of cell wall polymers is indispensable for insect defense.
In plant cells, cellulose microfibrils make up the basic skeleton of the rigid cell wall while hemicellulose and pectin fill the space between cellulose microfibrils. Cell walls constitute the first wall in plant defense against pathogens and cell wall polysaccharides, and their acetylation status greatly affects responses to pathogens. Many cell wall polysaccharides undergo extensive O-acetylation, imparting important physiochemical properties to the polymers. The O-acetylation of polysaccharides modifies plant cell walls by changing their physicochemical properties and, consequently, their structure and function. In this report, Sun et al. (10.1104/pp.20.00683) cloned a previously identified stunted growth mutant named sucrose uncoupled1 (sun1) in Arabidopsis (Arabidopsis thaliana). SUN1 encodes a member of the TRICHOME BIREFRINGEN-LIKE family, AtTBL37. TBL37 (SUN1) is highly expressed in fast-growing plant tissues and encodes a Golgi apparatus-localized protein that regulates secondary cell wall thickening and acetylation. In tbl37-1, jasmonate signaling and the expression of downstream chemical defense genes, are increased but, cell wall acetylation is reduced, resulting in stunted growth, collapsed xylem vessels, and increased susceptibility to herbivorous insects. A key transcription factor involved in jasmonate signaling, MYC2, binds to and induces AtTBL37 expression and also enhances cell wall acetylation. The mutant myc2-2 is as susceptible to insect feeding as tbl37-1. The overexpression of AtTBL37 in a myc2-2 background increases resistance to herbivory. These findings reveal that AtTBL37-mediated acetylation of cell wall polymers is indispensable for insect defense.