Make, Modify, Move: Multilayered Regulation of ONAC054 During ABA-Induced Leaf Senescence
‘Senescence’ originates from the Latin word senescere, to grow old. However, the process of leaf senescence is not simply the passive death of the leaf but instead represents an active and highly regulated process of nutrient remobilization from older leaves into developing parts of the plant to support further growth and reproduction. In crop plants an efficient senescence program is essential to maximize the carbohydrate and nutrient content of the seed, which ultimately impacts upon grain yield and quality. Post-harvest senescence of fruit and vegetables is also a significant economic issue, negatively affecting shelf-life and nutritional value. Aside from being a developmental process, senescence can also be induced prematurely as an adaptive response to environmental stress conditions.
Several transcription factor (TF) families have been implicated in the regulation of leaf senescence, including the bZIPs, NF-Ys, WRKYs, and NACs (Breeze et al., 2011). Many of these TFs are also known to be involved in phytohormone signalling pathways supporting the existence of a complex regulatory network underpinning leaf senescence. Here, Sakuraba et al. (2020) describe the mechanism of action of one such senescence-induced NAC TF in rice (Oryza sativa), ONAC054, which promotes ABA-induced leaf senescence.
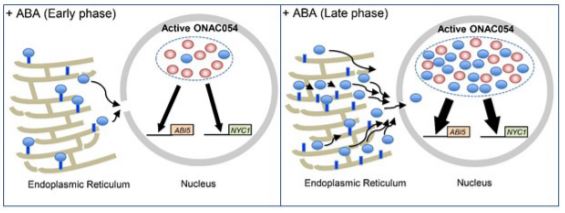
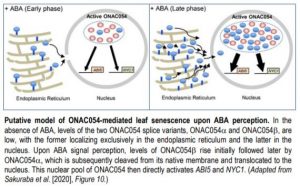 ONAC054 belongs to a subclass of NACs which are membrane-bound, frequently to the endoplasmic reticulum, by virtue of possessing a C-terminal transmembrane domain (TMD). Upon signal perception the TMD is proteolytically cleaved to release the active form of the TF which relocates to the nucleus (Kim et al., 2006). Sakuraba et al. (2020) demonstrated that ONAC054 (ONAC054a), is indeed dual-localized to the ER or nucleus depending on the presence or absence of a specific TMD. Moreover, they identified an alternative splice variant, ONAC054b, in which a premature stop codon effectively eliminates the TMD resulting in the truncated protein being solely observed in the nucleus.
ONAC054 belongs to a subclass of NACs which are membrane-bound, frequently to the endoplasmic reticulum, by virtue of possessing a C-terminal transmembrane domain (TMD). Upon signal perception the TMD is proteolytically cleaved to release the active form of the TF which relocates to the nucleus (Kim et al., 2006). Sakuraba et al. (2020) demonstrated that ONAC054 (ONAC054a), is indeed dual-localized to the ER or nucleus depending on the presence or absence of a specific TMD. Moreover, they identified an alternative splice variant, ONAC054b, in which a premature stop codon effectively eliminates the TMD resulting in the truncated protein being solely observed in the nucleus.
Loss-of-function onac054 mutants exhibited a functional stay-green phenotype. However, whilst the concomitant increases in photosynthetic capacity can be associated with higher crop yield, in this case the mutants were observed to have a reduced fertility, which negated any such benefits. Microarray analysis of senescent leaves of the onac054 mutant revealed that several genes associated with chlorophyll degradation (e.g. STAY-GREEN1 [SGR] and NON-YELLOW COLORING1 [NYC1]), and with both ABA biosynthesis and signalling (e.g. ABA-RESPONSIVE ELEMENT BINDING FACTOR4 [ABF4] and ABA-INSENSITIVE [ABI5]) were mis-regulated in comparison to wild-type. Subsequent chromatin immunoprecipitation and protoplast transactivation assays confirmed that a subset of these putative downstream targets, namely ABI5 and NYC1, were directly regulated by ONAC054 through the binding of the TF to a defined promotor motif. Consequently, ONAC054, in concert with other ABA-responsive NAC TFs, regulates the initial stages of chlorophyll degradation through the activation of the chlorophyll b reductase, NYC1.
Finally, the authors attempted to elucidate the precise regulatory roles of the two alternative splice variants in ABA-induced senescence. Whilst the transcript levels of both ONAC054a and ONAC054b increased upon ABA treatment, ONAC054b was up-regulated more rapidly and to a greater extent, potentially through post-transcriptional regulation of the 3’ untranslated region. Since ONAC054b does not require post-translational cleavage for activation, this suggests that this splice variant may be involved in early transcriptional responses to increases in ABA concentrations, with ONAC054a transcription, processing and nuclear import occurring later following more prolonged stimulation.
In summary, the membrane-bound TF ONAC054 is a key activator of ABA-induced leaf senescence, the activity of which is precisely controlled at the transcriptional, post-transcriptional and post-translational levels. Such multilayer regulation offers multiple points of entry for potential manipulation, with the ultimate aim to develop an improved stay-green line offering higher grain yields. One possible strategy could be to ‘lock’ the TF on its native membrane and thus prevent its nuclear relocalization, a tactic employed by pathogen virulence proteins (McLellan et al., 2013).
Emily Breeze
School of Life Sciences, University of Warwick, Coventry, UK
ORCID ID: 0000-0001-5383-5448
REFERENCES
Breeze, E. et al. (2011). High-resolution temporal profiling of transcripts during Arabidopsis leaf senescence reveals a distinct chronology of processes and regulation. Plant Cell 23: 873–894.
Kim, Y.S., Kim, S.G., Park, J.E., Park, H.Y., Lim, M.H., Chua, N.H., and Park, C.M. (2006). A Membrane-Bound NAC Transcription Factor Regulates Cell Division in Arabidopsis. THE PLANT CELL ONLINE 18: 3132–3144.
McLellan, H., Boevink, P.C., Armstrong, M.R., Pritchard, L., Gomez, S., Morales, J., Whisson, S.C., Beynon, J.L., and Birch, P.R.J. (2013). An RxLR effector from Phytophthora infestans prevents re-localisation of two plant NAC transcription factors from the endoplasmic reticulum to the nucleus. PLoS Pathog 9: e1003670.
Sakuraba, Y., Kim, D., Han, S.-H., Kim, S.-H., Piao, W., Yanagisawa, S., An, G., and Paek, N.-C. (2020). Multilayered regulation of membrane-bound ONAC054 is essential for abscisic acid-induced leaf senescence in rice. The Plant Cell https://doi.org/10.1105/tpc.19.00569.