A Tapetal Boric Acid Channel Involved in Pollen Cell Wall Formation
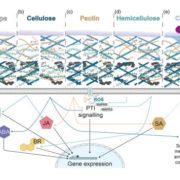
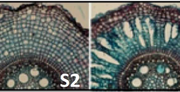
 Boron (B) is an essential plant micronutrient that plays a major role in cell wall structure and function by providing cross-linking of the rhamnogalacturonan II (RG-II) pectin component of the cell wall. Under normal pH conditions, B is found principally as boric acid. Although boric acid permeates lipid bilayers, it exhibits limited systemic mobility within most plant species, and this can contribute to B deficiency under low boric acid conditions. However, since boric acid is toxic at high concentrations, the maintenance of its cellular concentration is also critical. Consequently, boric acid uptake and homeostasis need to be tightly regulated. Among the membrane channels that facilitate boric acid uptake are the type II nodulin intrinsic protein (NIP) subfamily of aquaporin-like proteins. Arabidopsis thaliana possesses three NIP II genes (NIP5;1, NIP6;1, and NIP7;1) that show distinct tissue expression profiles (predominantly expressed in roots, stem nodes, and developing flowers, respectively). Routray et al. (10.1104/pp.18.00604) show that purified and reconstituted NIP7;1 is a boric acid facilitator. By using native promoter-reporter fusions, the authors show that NIP7;1 is predominantly expressed in anthers of young flowers during a narrow developmental window, with protein accumulation solely within tapetum cells where it is localized to the plasma membrane. The tapetum is essential for supplying materials to the pollen microspore cell wall. The authors report that under limiting boric acid conditions, loss-of-function T-DNA mutants (nip7;1-1 and nip7;1-2) show reduced fertility including shorter silique and an increase in aborted seeds compared to the wild type. Under these conditions, nip7;1 mutant pollen grains show morphological defects, increased aggregation, defective exine cell wall formation, reduced germination frequency, and decreased viability. Thus, it appears that NIP7;1 serves as a gated boric acid channel in developing anthers where it aids in the uptake of boric acid by tapetal cells.
Boron (B) is an essential plant micronutrient that plays a major role in cell wall structure and function by providing cross-linking of the rhamnogalacturonan II (RG-II) pectin component of the cell wall. Under normal pH conditions, B is found principally as boric acid. Although boric acid permeates lipid bilayers, it exhibits limited systemic mobility within most plant species, and this can contribute to B deficiency under low boric acid conditions. However, since boric acid is toxic at high concentrations, the maintenance of its cellular concentration is also critical. Consequently, boric acid uptake and homeostasis need to be tightly regulated. Among the membrane channels that facilitate boric acid uptake are the type II nodulin intrinsic protein (NIP) subfamily of aquaporin-like proteins. Arabidopsis thaliana possesses three NIP II genes (NIP5;1, NIP6;1, and NIP7;1) that show distinct tissue expression profiles (predominantly expressed in roots, stem nodes, and developing flowers, respectively). Routray et al. (10.1104/pp.18.00604) show that purified and reconstituted NIP7;1 is a boric acid facilitator. By using native promoter-reporter fusions, the authors show that NIP7;1 is predominantly expressed in anthers of young flowers during a narrow developmental window, with protein accumulation solely within tapetum cells where it is localized to the plasma membrane. The tapetum is essential for supplying materials to the pollen microspore cell wall. The authors report that under limiting boric acid conditions, loss-of-function T-DNA mutants (nip7;1-1 and nip7;1-2) show reduced fertility including shorter silique and an increase in aborted seeds compared to the wild type. Under these conditions, nip7;1 mutant pollen grains show morphological defects, increased aggregation, defective exine cell wall formation, reduced germination frequency, and decreased viability. Thus, it appears that NIP7;1 serves as a gated boric acid channel in developing anthers where it aids in the uptake of boric acid by tapetal cells.