Shedding New Light on the Ancient Process of Photosynthesis in Cyanobacteria
All photosynthetic organisms, from cyanobacteria to flowering plants, must continuously adjust to changing light conditions to maximize photosynthetic efficiency while protecting their delicate photochemical centers. During photosynthesis, light energy is preferentially captured by photosystem I (PSI) or PSII, depending on light levels and quality. The specific illumination of either pigment–protein complex creates an energy imbalance, leading to the reduction or oxidation of the intersystem electron transport chain. This energy imbalance is quickly restored via a mechanism known as state transitions (reviewed in Minagawa, 2011). State transitions are triggered by changes in the redox state of the membrane-soluble plastoquinone (PQ) pool located in the electron transport chain between PSII and the cytochrome b6f (cyt b6f) complex. In plants and green algae, these changes in redox potential are sensed by cyt b6f, which interacts with a specific kinase that alters the phosphorylation status of light-harvesting complex II (LHCII). This phosphorylation allows LHCII to shuttle between PSI and PSII, thereby redistributing excitation energy between the photosystems as needed. Such an arrangement enables the organism to efficiently utilize absorbed light to construct organic molecules from carbon dioxide and water, which helps make life on Earth possible.
While much is known about state transitions in plants, how they evolved is unclear, in part due to our poor understanding of this process in cyanobacteria, the first oxygen-evolving photosynthetic organisms. Calzadilla (2019) recently discovered that surprisingly, state transitions in the model cyanobacteria Synechocystis PCC 6803 and Synechococcus elongatus occur in a manner quite different from that of plants and green algae. First, a process in PSII associated with fluorescence quenching independent of PSI plays an important role in cyanobacterial state transitions. This process appears to be more relevant than fluorescence changes coming from the cyanobacterial extramembrane antenna known as the phycobilisome, at least in S. elongatus. This was probed using various hyperosmotic buffers that inhibit the movement of phycobilisomes, as well as processes occurring in the membrane photosystems, through analysis of chlorophyll fluorescence emission spectra obtained at 77 Kelvin (K). At this temperature (−196.15°C), the fluorescence of both photosystems is visible, while at room temperature, only PSII-related fluorescence is observed. The authors monitored changes in 77 K fluorescence spectra after inducing state transitions by exposing dark-adapted (State II) cells to light (State I).
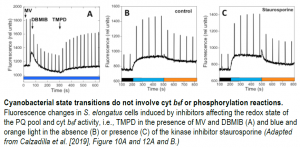 Second, by tracing fluorescence kinetics at room temperature with a PAM (pulse-amplitude modulation) fluorometer in cyanobacteria treated with various chemicals affecting the redox state of the PQ pool and cyt b6f activity, the authors demonstrated that the cyt b6f complex is not involved in state transitions in S. elongatus or Synechocystis (see figure). It appears that no specific phosphorylation reaction participates in cyanobacterial state transitions either. When 21 dark-adapted protein kinase and phosphatase mutants were successively illuminated with blue and orange light (to induce state transitions), they all exhibited similar fluorescence kinetics to those of the wild type. This finding was confirmed using kinase and phosphatase inhibitors (see figure).
Second, by tracing fluorescence kinetics at room temperature with a PAM (pulse-amplitude modulation) fluorometer in cyanobacteria treated with various chemicals affecting the redox state of the PQ pool and cyt b6f activity, the authors demonstrated that the cyt b6f complex is not involved in state transitions in S. elongatus or Synechocystis (see figure). It appears that no specific phosphorylation reaction participates in cyanobacterial state transitions either. When 21 dark-adapted protein kinase and phosphatase mutants were successively illuminated with blue and orange light (to induce state transitions), they all exhibited similar fluorescence kinetics to those of the wild type. This finding was confirmed using kinase and phosphatase inhibitors (see figure).
While much remains to be learned about the signal transduction pathway underlying state transitions in cyanobacteria, we now know that this pathway is completely different from that of plants and green algae. These findings suggest that the cyt b6f-dependent (and phosphorylation-dependent) state transition mechanism of plants and algae evolved after the endosymbiotic uptake of the cyanobacterium that gave rise to chloroplasts. Further analysis of state transitions in cyanobacteria would shed light on the evolution of photosynthesis, a process so crucial for life on Earth.
REFERENCES
Calzadilla, P.I., Zhan, J., Sétif, P., Lemaire, C., Solymosi, D., Battchikova, N., Wang, Q., and Kirilovsky, D. (2019). The cytochrome b6f complex is not involved in cyanobacterial state transitions. Plant Cell 31: https://doi:10.1015/tpc18.00916.
Minagawa, J. (2011). State transitions—the molecular remodeling of photosynthetic supercomplexes that controls energy flow in the chloroplast. Biochim. Biophys. Acta 1807: 897-905.



