Good Fats, Bad Fats: Phosphoinositide Species Differentially Localize to Plant-Pathogen Interfaces and Influence Disease Progression
Many filamentous pathogens invade living plant cells with specialized intracellular infection structures (haustoria) that promote microbial growth. Cytological studies demonstrate that the haustoria of fungal and oomycete pathogens are separated from host cell cytoplasm by a highly differentiated and de novo-formed extra-haustorial membrane (EHM). The differentiated EHM that surrounds invading haustoria, or the differentiated membrane separating host cells from the extra-invasive hyphae of non-haustorial intracellular pathogens, recruits a distinct set of proteins and cellular trafficking machinery in comparison to the host cell plasma membrane (Yi and Valent, 2013; Bozkurt and Kamoun, 2020).
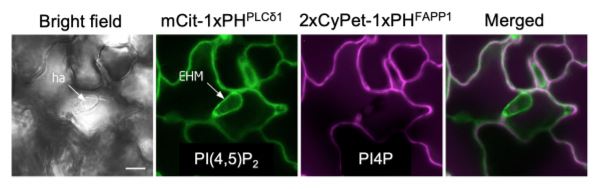
 Lipids are fundamental components of cellular membranes and the abundance of distinct lipid species often identifies specific sub-compartments within a given cell. In particular, the anionic phosphoinositide family of lipids contributes to the differentiation of endomembrane compartments to regulate cell patterning and direct polarized trafficking (Noack and Jaillais, 2017). To better understand the role of membrane lipids at plant-pathogen interfaces, Qin et al. (2020) surveyed phosphoinositide species present at the EHM during infection in Arabidopsis thaliana leaves. Using genetically-encoded phosphoinositide biosensor lines, the authors uncovered a robust and specific accumulation of the membrane lipid PI(4,5)P2 at the EHM surrounding Erysiphe cichoracearum (see Figure) or Albugo candida haustoria. This was further confirmed by phosphoinositide immunolocalization studies and co-localization experiments with known EHM markers such as the RPW8.2 (RESISTANCE TO POWDERY MILDEW 8.2) resistance protein. By contrast, the phosphoinositides PI4P and PI3P were both excluded from the EHM.
Lipids are fundamental components of cellular membranes and the abundance of distinct lipid species often identifies specific sub-compartments within a given cell. In particular, the anionic phosphoinositide family of lipids contributes to the differentiation of endomembrane compartments to regulate cell patterning and direct polarized trafficking (Noack and Jaillais, 2017). To better understand the role of membrane lipids at plant-pathogen interfaces, Qin et al. (2020) surveyed phosphoinositide species present at the EHM during infection in Arabidopsis thaliana leaves. Using genetically-encoded phosphoinositide biosensor lines, the authors uncovered a robust and specific accumulation of the membrane lipid PI(4,5)P2 at the EHM surrounding Erysiphe cichoracearum (see Figure) or Albugo candida haustoria. This was further confirmed by phosphoinositide immunolocalization studies and co-localization experiments with known EHM markers such as the RPW8.2 (RESISTANCE TO POWDERY MILDEW 8.2) resistance protein. By contrast, the phosphoinositides PI4P and PI3P were both excluded from the EHM.
To better understand how phosphoinositides are incorporated into the developing interface, the authors assessed the impact of pharmacological treatments on EHM localization during E. cichoracearum infection in the PI(4,5)P2 biosensor line. Treatments with latrunculin A (actin filament disruption), methyl-β-cyclodextrin (membrane sterol depletion), or wortmannin (phosphoinositide-kinase inhibitor) all strongly suppressed PI(4,5)P2 accumulation at E. cichoracearum haustoria, but neither oryzalin (microtubule disruption) nor brefeldin A (vesicular trafficking inhibitor) had any appreciable impact. This suggests that PI(4,5)P2 biogenesis and/or trafficking relies on an actin-dependent process involving membrane sterols and conserved phosphoinositide kinases.
The Arabidopsis thaliana genome encodes eleven PI4P 5-KINASE (PIP5K) isoforms thought to be responsible for PI(4,5)P2 biosynthesis (Mueller-Roeber and Pical, 2002). Through expression analysis and pip5k mutant phenotyping, the authors determined a redundant role for PIP5K1 and PIP5K2 in regulating PI(4,5)P2 biogenesis and function at the EHM. Compared to wild-type plants, pip5k1/pip5k2 double mutants displayed a significant reduction of PI(4,5)P2 levels throughout the cell and were more resistant to Erysiphe and Albugo infection. RNA-sequencing and phytohormone profiling of infected pip5k1/pip5k2 plants demonstrated that resistance was not correlated with the constitutive activation of classical defenses. Rather, pip5k double mutants displayed impaired actin filament assembly around invading pathogen structures. Collectively, Qin et al. describe a functional role for the PIP5K family in the establishment of a plant-pathogen interface that promotes disease progression, revealing PI(4,5)P2 as a novel susceptibility factor in plants.
Philip Carella
Sainsbury Laboratory, University of Cambridge, Cambridge
ORCID: 0000-0002-5467-7290
REFERENCES
Bozkurt TO and Kamoun S (2020) The plant–pathogen haustorial interface at a glance. J Cell Sci 133: jcs237958.
Mueller-Roeber B and Pical C (2002) Inositol Phospholipid Metabolism in Arabidopsis. Characterized and Putative Isoforms of Inositol Phospholipid Kinase and Phosphoinositide-Specific Phospholipase C. Plant Physiol. 130: 22–46.
Noack LC and Jaillais Y (2017) Precision targeting by phosphoinositides: how PIs direct endomembrane trafficking in plants. Current Opinion in Plant Biology 40: 22–33.
Qin L, Zhou Z, Li Q, Zhai C, Liu L, Quilichini TD, Gao P, Kessler SA, Jaillais Y, DatlaR, Peng G, Xiang D, Wei Y (2020) Specific recruitment of phosphoinositide species to the plant-pathogen interfacial membrane underlies arabidopsis susceptibility to fungal infection. Plant Cell Published March 2020 DOI: https://doi.org/10.1105/tpc.19.00970
Yi M and Valent B (2013) Communication Between Filamentous Pathogens and Plants at the Biotrophic Interface. Annu. Rev. Phytopathol. 51: 587–611.