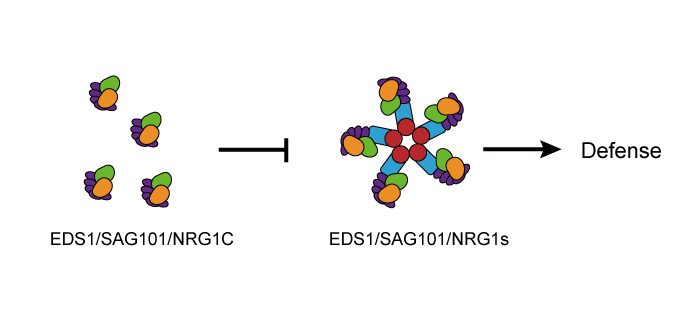
An N-terminally truncated NLR is functional
Background: Plants utilize sophisticated innate immune systems to fight against pathogens. Their genomes encode abundant immune receptors to detect the presence of pathogens and initiate defense responses. Nucleotide-binding leucine-rich repeat immune receptors (NLRs) constitute a major type of immune receptor, which are also present in animals. NLRs with pathogen recognition ability are sensor NLRs (sNLRs), whereas those required for sNLR-mediated defense are helper NLRs (hNLRs). NRG1 (N REQUIREMENT GENE1) family proteins are hNLRs that function with two lipase-like proteins, EDS1 (ENHANCED DISEASE SUSCEPTIBILITY1) and SAG101 (SENESCENCE-ASSOCIATED GENE 101), downstream of some sNLRs. Interestingly, N-terminally truncated NRG1C is clustered in tandem with full-length NRG1s in Brassicaceae genomes. However, little is known about the function of NRG1C in plant immunity.
Question: We asked whether N-terminally truncated NRG1C plays a role in plant immunity. If so, what is the molecular mechanism?
Findings: Using overexpression approaches, we found that NRG1C plays a negative role in plant immunity. Upon pathogen infection, NRG1C expression is induced. Furthermore, NRG1C antagonizes immunity mediated by the sNLRs that is dependent on full-length NRG1s. However, NRG1C does not affect the transcript or protein levels of full-length NRG1s, and it does not associate with them either. The interaction of NRG1C with EDS1 and SAG101 suggests that it might interfere with the EDS1-SAG101 complex, which signals together with full-length NRG1s.
Next steps: Besides NRG1C, other N-terminally truncated NLR (NL) genes are present in plant genomes. Additional investigations into the functions of such NL genes in plants will be of great interest to widen our understanding of plant NLRs and their regulation.
Zhongshou Wu, Lei Tian, Xueru Liu, Weijie Huang, Yuelin Zhang, Xin Li (2022). The N-terminally truncated helper NLR NRG1C antagonizes immunity mediated by its full-length neighbors NRG1A and NRG1B. https://doi.org/10.1093/plcell/koab285