The Butterfly Effect: Natural Variation of a Chloroplast tRNA-Modifying Enzyme Leads to Pleiotropic Developmental Defects in Rice
Translation of RNA information into protein is a fundamental process ensuring the production of functional proteins in cells. Transfer RNA (tRNA) is a vital component of the translation machinery as it delivers the correct amino acids to the elongating peptide chain based on codon-anticodon recognition. tRNAs are extensively modified at various sites, greatly contributing to translational accuracy and efficiency. To date, a number of tRNA-modifying enzymes have been identified to influence various developmental processes in distinct organisms (Björk G, 2005). However, much less is known about the functions of organelle tRNA-modifying enzymes in green plants (Karcher and Bock, 2009).
Liu et al. (2020) identified the functional gene PLEIOTROPIC DEVELOPMENTAL DEFECTS (PDD) encoding a tRNA-modifying enzyme located in the chloroplast. Near-isogenic line NIL-PDDOL plants were generated by backcrossing progenies repeatedly between African wild rice species as the donor parent and rice cultivar 187R as the recurrent parent. NIL-PDDOL exhibited severe pleiotropic developmental defects. At three-leaf stage, newly emerged leaves in NIL-PDDOL were albino and the photosynthetic pigment contents in NIL-PDDOL plants were much lower than in 187R. The ultrastructure of chloroplasts of NIL-PDDOL plants was also severely altered. NIL-PDDOL plants showed significantly reduced plant height, panicles length, and internode length, likely due to the smaller cell size compared to 187R.
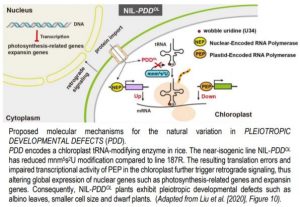 PDD encodes a tRNA-modifying GTPase belonging to the TrmE (transfer RNA modification E) family. The gene was preferentially expressed in green leaf tissues and PDD:eGFP fusion proteins co-localized with chloroplasts in rice protoplasts, indicating that PDD is a chloroplast-localized protein. The genetic variations in PDDOL caused two amino acid deletions at the N-terminus and five amino acid alterations along the entire protein region. Homodimerization ability of PDDOL was impaired and GTPase activity reduced relative to the wild-type 187R enzyme. The authors further demonstrated that PDD is involved in the 5-methylaminomethyl-(2-thio)uridine (mnm5s2U) modification of chloroplast tRNAs in rice.
PDD encodes a tRNA-modifying GTPase belonging to the TrmE (transfer RNA modification E) family. The gene was preferentially expressed in green leaf tissues and PDD:eGFP fusion proteins co-localized with chloroplasts in rice protoplasts, indicating that PDD is a chloroplast-localized protein. The genetic variations in PDDOL caused two amino acid deletions at the N-terminus and five amino acid alterations along the entire protein region. Homodimerization ability of PDDOL was impaired and GTPase activity reduced relative to the wild-type 187R enzyme. The authors further demonstrated that PDD is involved in the 5-methylaminomethyl-(2-thio)uridine (mnm5s2U) modification of chloroplast tRNAs in rice.
Given the importance of tRNA modification in ensuring the accuracy and efficiency of translation, the authors measured the abundance of several proteins. Chloroplast proteins associated with ribosome biogenesis and photosynthesis were greatly reduced in NIL-PDDOL plants. Further RNA-seq data revealed chloroplast-encoded genes with decreased mRNA levels were transcribed by plastid-encoding RNA polymerase (PEP), but genes with increased mRNA levels were transcribed by the nuclear-encoded RNA polymerase (NEP). Lastly, expression of ~4,000 nuclear genes was altered in NIL-PDDOL. The global expression of photosynthesis-related nuclear genes and expansin genes were repressed in NIL-PDDOL plants. The authors reasoned that the defective function of PDDOL results in decreased chloroplast tRNA modification, leading to translation errors in the chloroplast and impaired PEP activity, thereby abnormal chloroplast translation and transcription triggered retrograde signaling from the chloroplast to the nucleus (Chan et al., 2016), enhancing NEP activity and altering expression of nuclear genes (Figure).
Tian Zhang
Assistant Features Editor
ORCID: 0000-0002-0453-1621
Björk, G.R. and Hagervall, T.G. (2005). Transfer RNA Modification. EcoSal Plus. doi:10.1128/ecosalplus.4.6.2
Chan, K.X., Phua, S.Y., Crisp, P., McQuinn, R., and Pogson, B.J. (2016). Learning the languages of the chloroplast: retrograde signaling and beyond. Annu. Rev. Plant Biol. 67: 25–53.
Karcher, D. and Bock, R. (2009). Identification of the chloroplast adenosine-to-inosine tRNA editing enzyme. RNA 15: 1251–1257.
Liu, H. et al. (2020). A natural variation in PLEIOTROPIC DEVELOPMENTAL DEFECTS uncovers a crucial role for chloroplast tRNA modification in translation and plant development. Plant Cell DOI: https://doi.org/10.1105/tpc.19.00660.