Proteins that Function in Lipid Droplets
Kretzschmar et al. study the proteome of lipid droplets and identify a protein that functions in protein degradation during germination https://doi.org/10.1105/tpc.18.00276
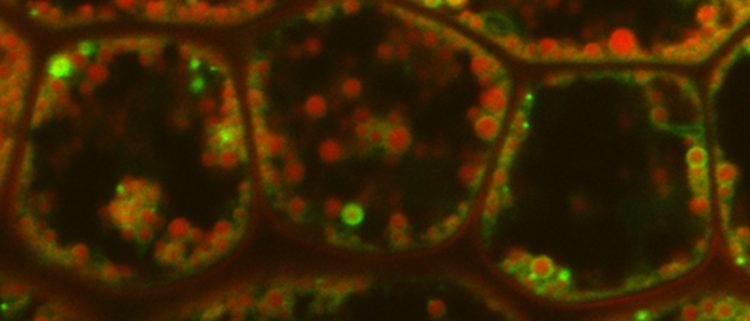
Background: Lipid droplets (LDs) are structures inside cells that store oil or fat, for example, in oil seeds. They were once considered plain storage organelles. Ongoing research in yeast and animal systems, however, has revealed that LDs are dynamic organelles with complex functions. In plants, new roles for LDs are emerging, too. Nevertheless, knowledge about the proteins embedded in the surface of plant LDs is limited. As our first model to study, we chose tobacco pollen tubes, which contain a high number of LDs. Pollen tubes are formed by pollen grains during plant reproduction, but can also form in growth medium.
Question: We first aimed to identify previously unknown LD-associated proteins. We then chose one of the newly identified LD proteins to study its role in LD biology.
Findings: We compared the protein composition—the proteome—of LDs isolated from tobacco pollen tubes to the total proteome of pollen tubes. Based on these data, we identified candidates that were highly enriched in the LD fractions. From this list, we chose several proteins and verified their LD localization by confocal laser scanning microscopy. Thereby, we identified three previously unknown LD proteins. We chose one of these proteins, named PUX10, for an in-depth characterization and also extended our work on this protein to the model system Arabidopsis thaliana. We discovered that PUX10 is involved in the degradation of other proteins sitting on the surface of LDs during seed germination, when LDs are broken down to fuel the germination process. PUX10 appears to do so by binding proteins that need to be degraded and also binding another protein, CDC48, known to be important for protein degradation. PUX10 functions as a bridge, or scaffold, between these proteins and brings them together.
Next steps: Our findings extend the plant LD proteome by three proteins and reveal a player involved in the regulation of the plant LD proteome. However, many open questions concerning various processes of LD biology remain. Therefore, the identification and characterization of further proteins involved in these processes is needed.
Franziska K. Kretzschmar, Laura A. Mengel, Anna O. Müller, Kerstin Schmitt, Katharina F. Blersch, Oliver Valerius, Gerhard H. Braus, Till Ischebeck. (2018). PUX10 Is a Lipid Droplet-Localized Scaffold Protein That Interacts with CELL DIVISION CYCLE48 and Is Involved in the Degradation of Lipid Droplet Proteins. Plant Cell 30: 2137-2160; DOI: https://doi.org/10.1105/tpc.18.00276.