A Role for Autophagy in Plant Lipid Homeostasis
Autophagy is the conserved eukaryotic mechanism by which cytoplasmic components such as macromolecular complexes, organelles, and cytosol are degraded in the lysosome or vacuole (Reggiori and Klionsky, 2013). Basal autophagy ensures that obsolete organelles and misfolded proteins are removed from the cytosol, whereas induced autophagy is an adaptive response to environmental stress. A high-throughput metabolomic, lipidomic, and proteomic analysis of Arabidopsis thaliana mutants lacking functional AUTOPHAGY-RELATED (ATG) genes showed that autophagy affects central metabolism under carbon starvation conditions (Avin-Wittenberg, 2015). While lipophagy (i.e., the autophagic degradation of lipid droplets) is known to play important roles in lipid turnover in yeast and mammals (van Zutphen et al., 2014 and Schulze et a., 2017), little is known about this process in plants.
To address this knowledge gap, Fan et al. (2019) examined the contribution of autophagy to lipid droplet homeostasis in Arabidopsis. Analysis of triacylglycerol (TAG) content revealed that disruption of basal autophagy (through mutation of ATG2 or ATG5) reduces TAG accumulation in seeds, seedlings, and leaves, disrupts TAG synthesis in mature and senescing leaves, and decreases the mobilization of fatty acids from membrane lipids to TAG. These observations suggest that basal autophagy contributes to organellar membrane lipid turnover via TAG synthesis. Studies of a series of higher-order mutants in which TAG artificially accumulates in the leaves demonstrated that basal autophagy influences both fatty acid biosynthesis and membrane lipid turnover, with the ER lipid assembly pathway having a greater role in this process than the chloroplast pathway.
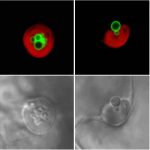
 To examine whether autophagy contributes to TAG breakdown in lipid droplets, the authors monitored the localization of lipid droplets and autophagic structures in leaves subjected to dark-induced starvation using transgenic lines overexpressing the lipid droplet marker OLE1-GFP and the autophagosome marker DsRed-ATG8e. Increased autophagic activity was detected after a 3-day exposure to darkness and, while lipid droplets and autophagosomes rarely colocalized under normal growth conditions, the autophagic structures frequently associated with lipid droplets after dark-induced starvation. Further analysis indicated that the OLE-GFP-coated lipid droplets were degraded in vacuoles during exposure to darkness, indicating that lipophagy is induced by this treatment. Using both fluorescence and immunoelectron microscopy techniques, the authors next demonstrated that lipid droplets are enclosed in the vacuoles of plants exposed to dark-induced starvation, and that the process morphologically resembles microlipophagy, the type of lipophagy described in yeast. Disruption of autophagy inhibited localization of lipid droplets to the vacuole and autophagic degradation.
To examine whether autophagy contributes to TAG breakdown in lipid droplets, the authors monitored the localization of lipid droplets and autophagic structures in leaves subjected to dark-induced starvation using transgenic lines overexpressing the lipid droplet marker OLE1-GFP and the autophagosome marker DsRed-ATG8e. Increased autophagic activity was detected after a 3-day exposure to darkness and, while lipid droplets and autophagosomes rarely colocalized under normal growth conditions, the autophagic structures frequently associated with lipid droplets after dark-induced starvation. Further analysis indicated that the OLE-GFP-coated lipid droplets were degraded in vacuoles during exposure to darkness, indicating that lipophagy is induced by this treatment. Using both fluorescence and immunoelectron microscopy techniques, the authors next demonstrated that lipid droplets are enclosed in the vacuoles of plants exposed to dark-induced starvation, and that the process morphologically resembles microlipophagy, the type of lipophagy described in yeast. Disruption of autophagy inhibited localization of lipid droplets to the vacuole and autophagic degradation.
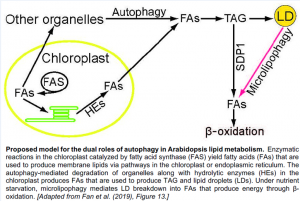
Based on these observations, the authors created a model representing the dual roles of autophagy in lipid droplet biogenesis and turnover (figure). Future studies will aim to refine the underlying mechanism and evaluate how this mechanism is regulated.
References
Avin-Wittenberg, T., Bajdzienko, K., Wittenberg, G., Alseekh, S., Tohge, T., Bock, R., Giavalisco, P., and Fernie, A.R. (2015). Global Analysis of the Role of Autophagy in Cellular Metabolism and Energy Homeostasis in Arabidopsis Seedlings under Carbon Starvation. Plant Cell 27:306–322.
Fan, J., Yu, L., and Xu, C. (2019). Dual Role for Autophagy in Lipid Metabolism in Arabidopsis. Plant Cell tpc.00170.2018; DOI: 10.1105/tpc.19.00170. (https://doi.org/10.1105/tpc.19.00170)
Reggiori, F. and Klionsky, D.J. (2013). Autophagic Processes in Yeast: Mechanism, Machinery and Regulation. Genetics 194:341–361.
Schulze R.J., Sathyanarayan A., and Mashek D.G. (2017). Breaking fat: The regulation and mechanisms of lipophagy. Biochim Biophys Acta Mol Cell Biol Lipids. 1862:1178–1187.
Van Zutphen, T., Todde, V., de Boer, R., Kreim, M., Hofbauer, H.F., Wolinski, H., Veenhuis, M., van der Klei, I.J., and Kohlwein, S.D. (2014). Lipid droplet autophagy in the yeast Saccharomyces cerevisiae. Mol Biol Cell. 25:290-301.