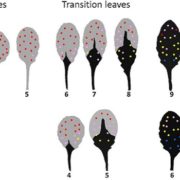
Annexins Facilitate Post-Phloem Sugar Transport
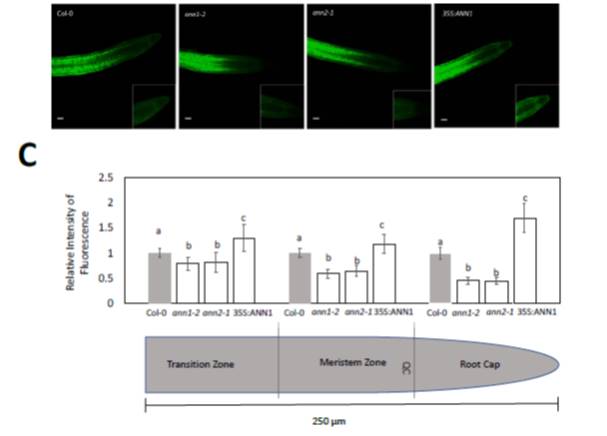
Because local sugar concentrations play a critical role in regulating root growth, it is important to learn more about the mechanisms that control sugar transport into the growing zones of the root. It has previously been proposed that once translocated sugar reaches the interface between conducting phloem and sink tissues in the root, it is unloaded into surrounding cells through plasmodesmata, whereupon it diffuses symplastically into cells of the root tip. The unloading of sugar into the elongation zone, mitotic zone, and root cap accommodates the high-energy demands of fast-growing regions in roots. Annexins are calcium-dependent membrane-binding proteins that play important roles in cell signaling. Annexin proteins have been identified in the phloem sap of several different plant species, and in Arabidopsis (Arabidopsis thaliana), the annexin AnnAt1 was found in phloem exudate of rosette leaves. In situ transcript localization studies have previously found that the annexin gene AnnAt1 is expressed in phloem and phloem parenchyma in Arabidopsis seedlings. However, the physiological importance of annexins in phloem remains undefined. Wang et al. (10.1104/pp.18.00713) describe the effect of knocking out two of the eight members of the annexin gene family in Arabidopsis, namely AnnAt1 and AnnAt2, on the diffusion of sugars from the unloading zone to root tips. They report that the inhibition of primary root growth and rootcap development in AnnAt1 and AnnAt2 knockout mutants is rescued by exogenous sucrose independently of its osmotic effects. Knocking out AnnAt1 and AnnAt2 also increases reactive oxygen species levels and callose accumulation in root tips and impairs post-phloem transport in root tips. Assuming that some of the accumulated callose is deposited in plasmodesmata, it would restrict plasmodesmatal sugar transport, and thus help explain why post-phloem transport of sugar to root tips is inhibited in mutants.